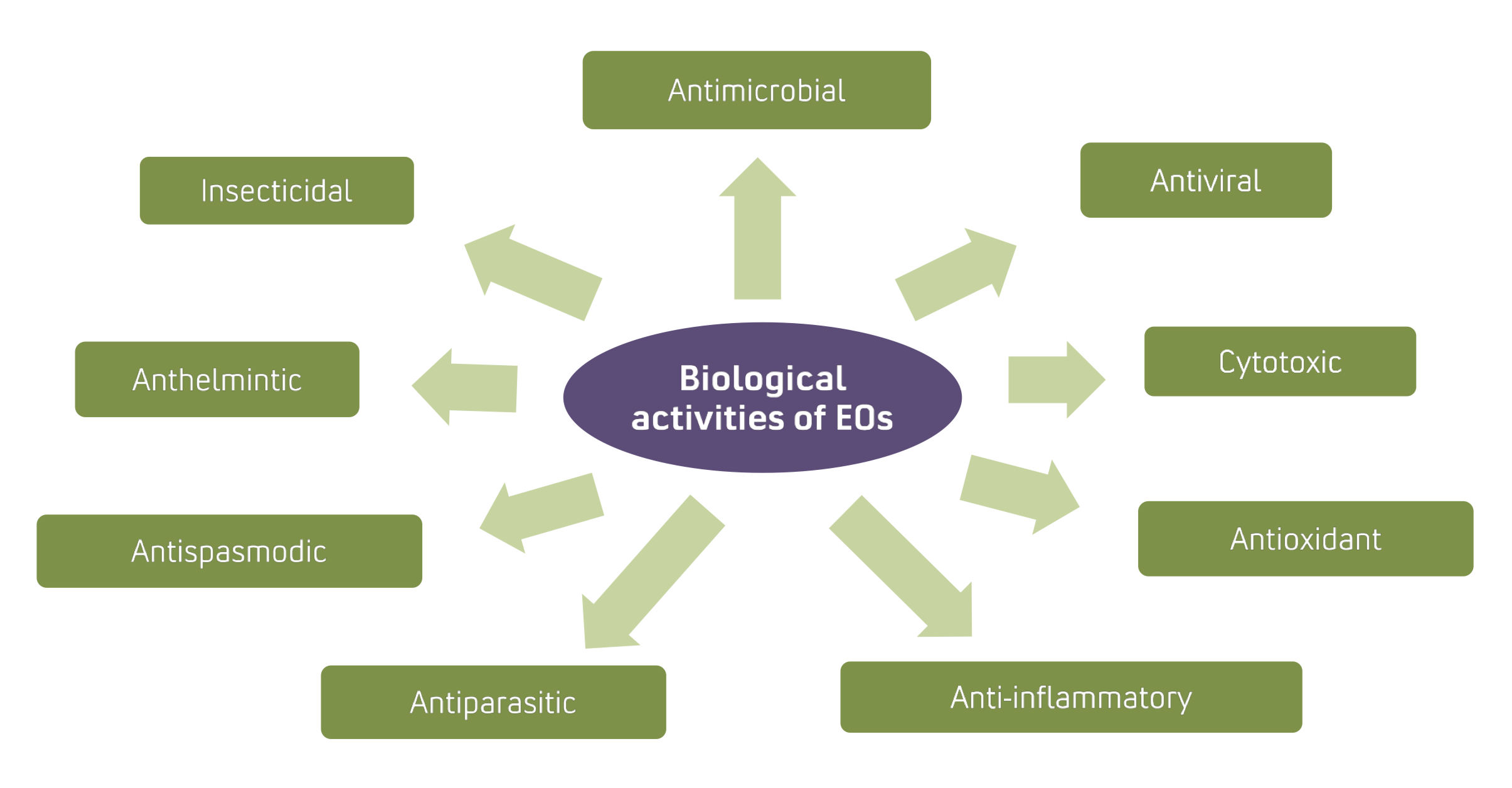
Essential oils are volatile, complex products of plants as secondary metabolites and include terpenes and their oxygenated derivatives, such as alcohols, aldehydes, esters, ketones, phenols and oxides.近些年来,从植物中提取的3000种基本油中,只有300种基本油在各个领域应用方面得到了广泛关注。[.]
Der Beitrag Essential oils are volatile, complex products of plants as secondary metabolites and include terpenes and their oxygenated derivatives, such as alcohols, aldehydes, esters, ketones, phenols and oxides.近些年来,从植物中提取的3000种基本油中,只有300种基本油在各个领域应用方面得到了广泛关注。在这次审查中,我们讨论与EO有关的主要生物活动,如杀菌剂、杀菌剂、杀菌剂、杀菌剂、杀菌剂、杀菌剂、杀菌剂、杀菌剂、杀虫剂、杀寄生物剂和细胞毒剂各种运送基本油路以及诸如高挥发性、低稳定性、渗透性、生物可用性、低水溶性、易氧化性、分解性、照片敏锐度和皮肤刺激等基本油问题也得到强调。此外,不同纳米封装系统建议解决上述问题的战略其中包括聚合纳米载体、脂基纳米载体和分子复合体相信纳米封装基础油将提高它们的治疗活动与交付量 。 
Science Vision – Volume 20, issue 2, pages 54–71 – 30 June 2020 https://doi.org/10.33493/scivis.20.02.01
Keywords: Biological activity, cytotoxic, antiviral, antiparasitic, nanocapsulation,Polymer-based nanocarriers, Nanocapsules, Nanoparticles, lipid-based nanocarriers, Lauroglycol® FCC, Capryol® 90, medium chain triglycerides, paraffin oil, 2-octyldodecanol, Miglyol® 812, Transcutol® HP, Labrafil Lipofile® WL 1349, Labrafac® PG, Polyvinyl alcohol, Solutol® HS15, polyglycerol methyl glucose distearate Pluronic® F68 (poloxamer 188), Pluronic® F127, Tween 20, Tween 40, Tween 80, Phosphatidylcholines, phosphatidylethanolamines, Gelucire® 50/13, Polysorbate20, Polysorbate80, Polyoxy60, castor oil, glycerine, PEG300, PEG400, Polyene glycol, Poloxamer
Der Beitrag Essential oils: a review on their salient biological activities and major delivery strategies erschien zuerst auf Pharma Excipients.

Current study evaluated the processability and characteristics of prills made of an active pharmaceutical ingredient/fatty acid (API/FA) suspension instead of previously studied API/FA solutions to enlarge the application field of prilling.Metformin盐化物(MET)和parecatamol(PAR)都被用作API模型使用,而药载效果(10-40%)和FA链 [.]
DerBeitrag Current study evaluated the processability and characteristics of prills made of an active pharmaceutical ingredient/fatty acid (API/FA) suspension instead of previously studied API/FA solutions to enlarge the application field of prilling. Metformin hydrochloride (MET) and paracetamol (PAR) were used as model APIs while both the effect of drug load (10 to 40%) and FA chain length (C14 to C22) were evaluated.API/FA悬浮可处理实验级定价设备,无热分解、喷嘴阻塞或沉积作用处理时间收集芯片面平滑(球度0.914)和粒子大小分别为++2.3毫米和2.4毫米,与药荷和/或FA链长无关体外放药评价显示,高药载量、高API水溶性和短FA链长度释放药量更快。 Solid状态分解通过XRD和Raman光谱显示,API和FA晶体化经热处理后通过prilling和存储期间维护相似性系数评价显示MET和PARprils在6个月存储25°C或40°C后稳定释放药量(f2>50). 
Der Beitrag Prilling of API/fatty acid suspensions: processability and characterisation erschien zuerst auf Pharma Excipients.

Oral drug delivery technology is mainly provided in the form of solid oral dosage forms (SODF) that have to be swallowed intact and move throughout the oro-esophageal system to release the drug content in the stomach or intestine.渐渐有证据表明某些疾病的吞化功能越来越普遍 [.]/p>Der Betrag Oral drug delivery technology is mainly provided in the form of solid oral dosage forms (SODF) that have to be swallowed intact and move throughout the oro-esophageal system to release the drug content in the stomach or intestine.越来越多的证据表明,在某些疾病、多发性和成熟度中,破损吞咽功能日益普遍,预测SODF奥氏滑翔法将非常有用。 第一阶段系统测量动聚合物表面所需的强度,即与脉冲层相接触,第二阶段则阻抗行为超出定义长度所得结果显示,综合滑动剖面视聚合膜测试而定。 
Der Beitrag Gliding performance of solid oral dosage form film coatings using an artificial mucous layer erschien zuerst auf Pharma Excipients.
Implication of Quantitative Selection of Each Excipient in Product Development Excipients' role in designing different dosage forms does not require any introduction.以上这些添加物与药理活性物质并发增加这些特性的主要目的是增加大数配方并传递所期望的[.]
Beitrag Excipients' role in designing different dosage forms does not require any introduction.以上这些添加物与药理活性物质并发增加这些特性的主要目的是增加大片配方并传递期望属性始发者像药物一样,需要验证和标准化下一章简单介绍不同用量表使用前接收者,包括固态用量表、液态用量表和半固态用量表计药先进配方领域的持续开发,我们还覆盖纳米化前接收者。 
Implication of Quantitative Selection of Each Excipient in Product Development
Der Beitrag Current Developments in Excipient Science – Implication of Quantitative Selection of Each Excipient in Product Development erschien zuerst auf Pharma Excipients.
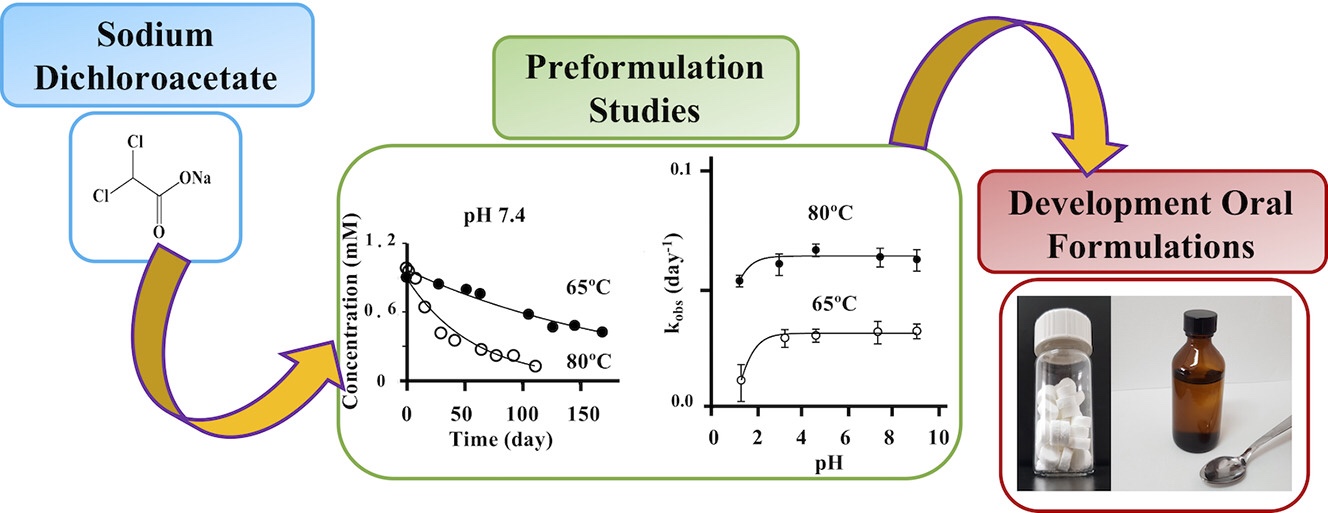
The purpose of this study was to develop liquid and solid paediatric formulations of sodium dichloroacetate (DCA) for the treatment of congenital lactic acidosis (CLA).在这次实验中,对活性分子进行了预编程研究以确定药物物理化学特性-二氯乙酸/'药性预编程研究'#/p>Beitrag The purpose of this study was to develop liquid and solid paediatric formulations of sodium dichloroacetate (DCA) for the treatment of congenital lactic acidosis (CLA).在这次工作上,对活性分子进行了预编程研究,以查明药物与剂量表设计及其制造过程相关联的物理化学属性。TGA和DSC分析显示DA钠非常染色HPLC和NMR分析显示,在所研究的所有pH值中,25和40摄氏度水解大都稳定基于这些结果,DA钠配制成可口化解法,含糖化物、粘度增强器和受CLA影响的儿科病人所容忍的香味前端开发液配方三个月后化学稳定在25摄氏4摄氏度使用稳定性测试显示一个月内没有化学降解和微生物污染DA钠口服片由仿制技术制成,作为一种替代和实用配方,比液型更容易为护理者管理技术解析(欧洲药典报告)显示口服片片快速分解水分3分钟25摄氏度,因此归为口服分解片片预写性研究提供一套参数,可据此进行详细的配方设计。Formulation studies showed that the developed dosage forms achieved adequate stability, producibility and patient acceptability. More on the pediatric formulation Der Beitrag Pharmaceutical preformulation studies and paediatric oral formulations of sodium dichloroacetate erschien zuerst auf Pharma Excipients.
