Purpose: Gums and polymers can be found abundantly in the nature from various plants, microbial and marine sources.它们的物理和功能特征可按产品开发应用定制研究的目的是确定从植物源获取自然口香糖开发gel eschienzuft Purpose: Gums and polymers can be found abundantly in the nature from various plants, microbial and marine sources.它们的物理和功能特征可按产品开发应用定制研究的目的是确定从植物源开发凝胶配方并与商业用聚合物Viz比较Carbopol 934、CorbeyMetCellulose、Xanthan、Sodium Alginite.br/ > Methods: Gel配方粘合粘合性 并选用其他口香糖 并更接近合成对口像 Carbopol 934和Sudium algate自然口香糖提供范围相对较广的特征和生态友好方法,增强配方开发应用性本研究工作理顺天然口香糖在开发药用量表、整形产品和养分配方方面的重要性。 
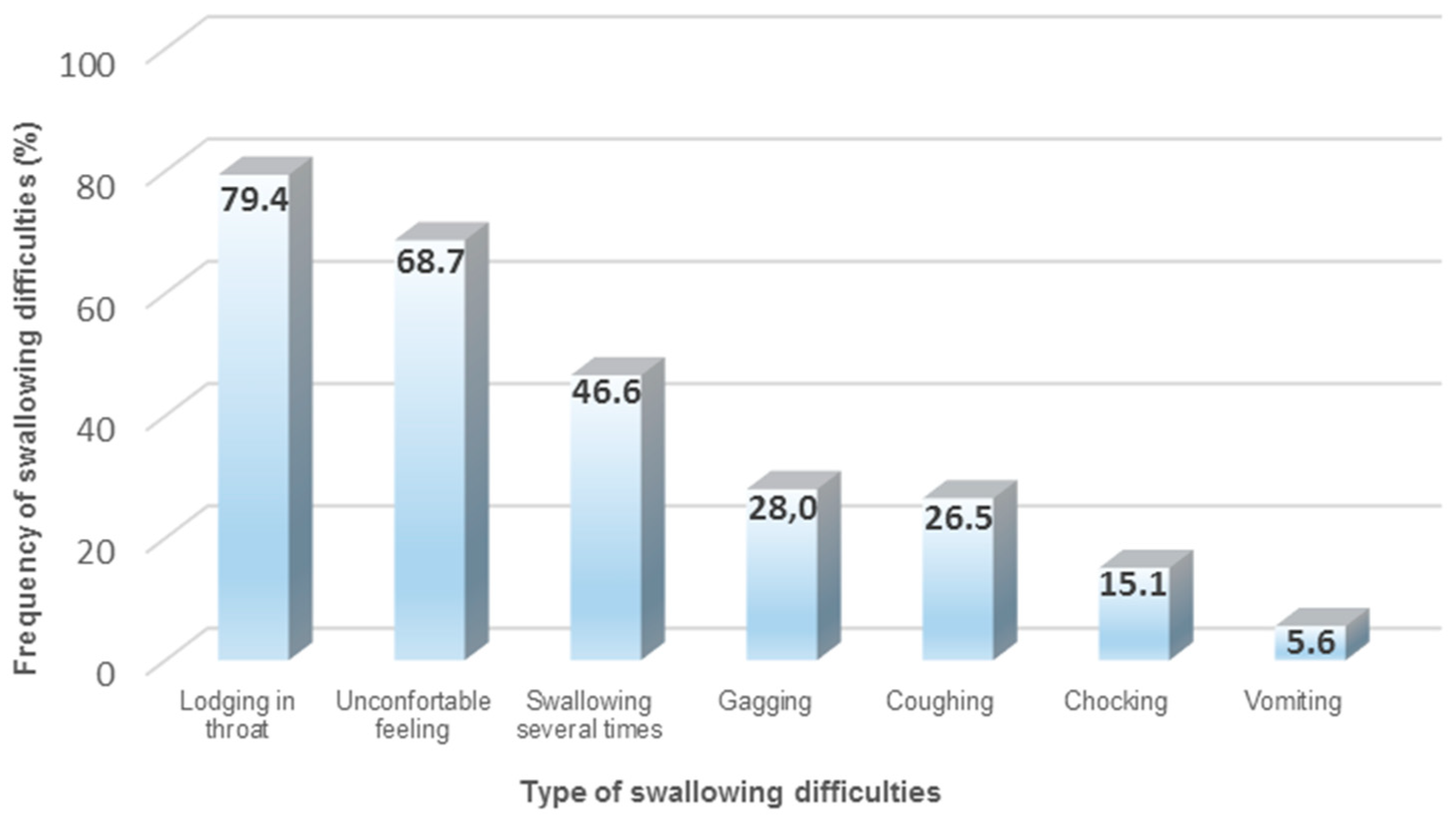
Oral drug administration provided as solid oral dosage forms (SODF) remains the major route of drug therapy in primary and secondary care.有明显证据表明,老年病人口中吞吐问题(例如Disphiga)越来越多,特别是考虑到多发性、虚弱和多药化病人时更是如此。Swallewing缺陷存有 [.]
Der Betrag Oral drug administration provided as solid oral dosage forms (SODF) remains the major route of drug therapy in primary and secondary care.有明显证据表明,老年病人口中吞吐问题(例如Disphiga)越来越多,特别是考虑到多发性、虚弱和多药化病人时更是如此。Swallowing缺陷对SODF管理有负面影响,导致加入率差和不适当的修改(例如压碎和拆分)。多年来提出了不同的策略,以便通过使用传统管理技术或吞食辅助装置增强SODF吞存经验尽管如此,新配方设计必须通过实施以病人为中心的方法加以考虑,以便有效改善老年病人对SODF的管理与适当的SODF规模缩放并发新片 
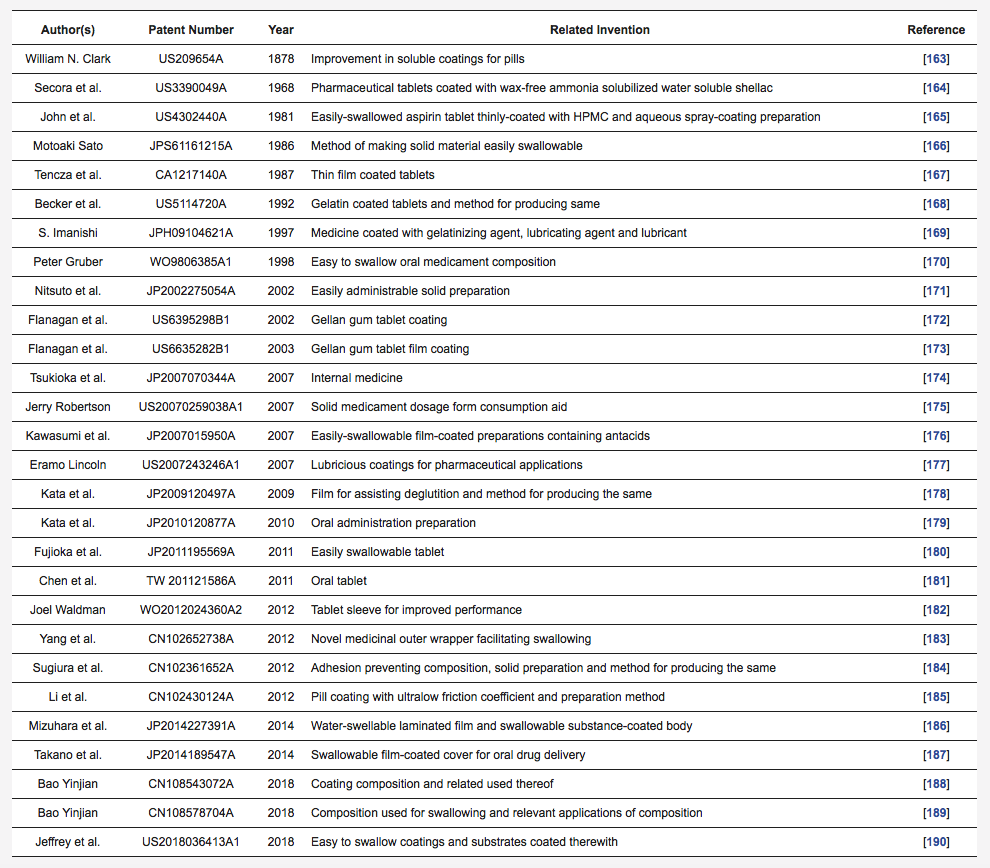
Table 7: Patents addressing new coating materials to enhance swallowability of SODF.
Der Beitrag Better Medicines for Older Patients: Considerations between Patient Characteristics and Solid Oral Dosage Form Designs to Improve Swallowing Experience erschien zuerst auf Pharma Excipients.

The objective of this study is to use a carbohydrate polymer deacetylated gellan gum (DGG) as matrix to design nanocrystals based intranasal in situ gel (IG) for nose-to -brain delivery of drug.HAR-NC模型药用同质喷雾技术编译HAR-NC解析并组成 [.]
Der Beitrag

The objective of this study is to use a carbohydrate polymer deacetylated gellan gum (DGG) as matrix to design nanocrystals based intranasal in situ gel (IG) for nose-to -brain delivery of drug.HAR-NC模型药用同质喷雾技术编译HAR-NC重新分布于dGG解决方案中并组成ionti晶状HAR在同化和喷雾干燥期间没有变化HAR-NC-IG和0.5%DGG显示精良现场凝聚能力、水保留属性和体外释放行为脑内HAR-NC-IG生物可用性比口口服HAR-NC高25倍,这可归结为HAR-NC和DGG生物沉积属性由鼻液触发的纳米化效果HAR-NC-IG可显著抑制乙酰胆碱酯酶表达方式并增加脑中乙酰胆碱化内容比引用配方内容(p < 0.01)dGG基础纳米晶体凝胶是一个大有前途的载体,可流出易解药,可延长宿期并增加脑中易解药的生物可用性。
/ahrefss/www.sciencedent.com/science/abs/pi/S0378517320311674国际药理学杂志,2020年https://doi.org/10.1016/j.jpharm.20120182.Der Beitrag

Plant-derived phenolic compounds have multiple positive health effects for humans attributed to their antioxidative, anti-inflammatory, and antitumor properties, etc.这些效果在很大程度上取决于生物机体中的生物可用性生物存取性,并因此生物存取性在很大程度上取决于生物体结构与形式,例如通过复合s/

Plant-derived phenolic compounds have multiple positive health effects for humans attributed to their antioxidative, anti-inflammatory, and antitumor properties, etc.这些效果在很大程度上取决于生物机体中的生物可用性生物存取性并因此生物可用性在很大程度上取决于生物体所输入的结构和形式,例如通过复合食物矩阵或净化分离倍数复合体与食物中或消化期间的其他大型分子(蛋白类、脂肪类、饮食纤维类、多功能类)发生交互作用,极大地影响生物接触生物机体,但由于倍数复合体机制复杂,尚未充分检测此区模拟胃消化法是常用体外测试之一,用于评估苯丙化合物生物存取性封装法可积极影响生物可获取性与生物可用性,因为它能确保
Check the encapsulation materials and methods in the following tables as of this publication
Encapsulation of phenolic acids and stilbenes
| Core Material | Wall Material | Encapsulation Method |
|---|---|---|
| ferulic acid | chitosan-tripolyphosphate pentasodium | ionic gelation |
| ferulic acid | poly-D,L-lactide-co-glycolide (PLGA) | double emulsion |
| caffeic acid | poly-D,L-lactide-co-glycolide (PLGA) | emulsion |
| syringic acid | D-Alpha tocopheryl polyethylene glycol 1000 succinate (TPGS) | thin-film dispersion |
| trans-resveratrol | zein | electrospraying |
| trans-resveratrol | poly-D,L-lactide-co-glycolide (PLGA) | precipitation |
Encapsulation of flavonoids
| Flavonoid Category | Core Material | Wall Material | Encapsulation Method |
|---|---|---|---|
| flavanols | quercetin | chitosan | ionic gelation |
| flavanols | quercetin | poly(lactic-co-glycolic acid) (PLGA) | emulsion diffusion evaporation |
| flavanols | quercetin | soluplus micelles | film dispersion |
| flavanols | quercetin | linseed oil, GMS, P6, Tween 80, 1,1-propylene glycol | high pressure homogenization |
| flavanols | quercetin | poly-D,L-lactide (PLA) | solvent evaporation |
| flavanols | quercetin | glycerol monostearate (GMS), medium chaintriglycerides (MCT), soy lecithin | emulsifying and solidifying |
| flavanols | quercetin | zein, 2-hydroxypropyl-β-cyclodextrin | spray-drying |
| flavanols | quercetin | casein, 2-hydroxypropyl-β-cyclodextrin | coacervation |
| flavanols | quercetin | poly(lactic-co-glycolic acid) (PLGA) | solvent displacement |
| flavanols | quercetin | ethylcellulose | precipitation |
| flavanols | quercetin | soy lecithin, glyceryl tridecanoate, glyceryl tripalmitate, vitamin E acetate, Kolliphor HS15 | phase inversion |
| flavanols | quercetin | (β-CD)-dodecylcarbonate | freeze-drying |
| flavanols | kaempferol | chitosan, sodium tripolyphosphate | ionic gelation |
| flavanols | kaempferol | lecithin–chitosan | electrostatic self-assembly |
| flavanols | fisetin | DOPC, cholesterol, DODA-PEG2000 | liposomes |
| flavanols | fisetin | PLGA (poly-lactide-co-glycolic acid), HPβCD (hydroxyl propyl beta cyclodextrin) | emulsion, freeze drying |
| flavones | tangeretin | zein | emulsion |
| flavones | apigenin | soybean oil, Tween 80 | in vitro digestion, in vivo pharmacokinetics |
| flavones | rutin | chitosan | ionic gelation |
| flavanones | naringenin | phospholipid, cholesterol, sodium cholate, and isopropyl myristate | liposomes by thin-film dispersion |
| flavan-3-ols | epigallocatechin gallate (EGCG) | gum arabic, maltodextrin | spray drying |
| flavan-3-ols | epigallocatechin gallate (EGCG) | chitosan-tripolyphosphate | freeze-drying |
| flavan-3-ols | catechin hydrate | phosphatidylcholine (PC) | liposomes |
| flavan-3-ols | catechin hydrate | horse chestnut, water chestnut and lotus stem starch | freeze drying |
| flavan-3-ols | green tea catechins | soy protein | emulsion |
| flavan-3-ols | green tea catechins | vitamin C and xylitol, γ-cyclodextrin and hydroxypropylmethyl cellulose phthalate | film-forming |
| flavan-3-ols | green tea catechins | hydroxypropyl methyl cellulose phthalate | coating |
| flavan-3-ols | tea catechins | corn oil and polysorbate 80 | emulsion |
| isoflavones | daidzein | phospholipid | film-homogenization |
| isoflavones | genistein | Soluplus® and Vitamin E d-α-Tocopheryl polyethylene glycol 1000 succinate (TPGS) | organic solvent evaporation |
Encapsulation of anthocyanins
| Core Material* | Wall Material | Encapsulation Method |
|---|---|---|
| blackberry purees | β-cyclodextrin | molecular inclusion |
| saffron anthocyanins | β-glucan and β-cyclodextrin | spray drying |
| Vaccinium ashei extracts | whey protein isolate | spray drying |
| Bryophyllum pinnatumextract | β-cyclodextrin | emulsion |
| bran extract | maltodextrin, gum arabic, whey protein isolate | spray drying |
| bran extract | alginate-whey protein isolate | ionic gelation |
| sour cherries skins extract | whey proteins isolate | freeze-drying |
| bilberry extract | whey protein, citrus pectin | emulsification and thermal gelation |
| anthocyanins standards mixture | cyclodextrins | freeze-drying |
| anthocyanins standards mixture | chitosan hydrochloride, carboxymethyl chitosan, β-Lactoglobulin | ionic gelation |
| bilberry extract | pectin amide | extrusion |
| bilberry extract | pectin amide with an additional shellac coating | emulsification/heat gelation |
| bilberry extract | whey proteins | spray drying |
| black carrot extract | polycaprolactone | double emulsion |
| black carrot extract | cholesterol and non-ionic surfactant (Tween 20) | niosome method |
| mulberry-extracted anthocyanin | alginate/chitosan | spray drying and external gelation |
| red pepper waste | whey protein | spray drying and freeze-drying |
| bilberry extract | whey protein isolate | gelation |
* Source of anthocyanins
Keywords: bioaccessibility!模拟胃肠消化目标交付控制释放封装技巧coating materials
Der Beitrag Role of the Encapsulation in Bioavailability of Phenolic Compounds erschien zuerst auf Pharma Excipients.

Rheumatoid arthritis (RA) is a chronic inflammatory disease characterized by joint synovial inflammation, as well as cartilage and bone tissue destruction.当前处理RA策略可减少联合炎症,但处理选项仍代表稳定问题,因为它们不足并快速清除数个药服系统 [.]
Der Beltrag Rheumatoid arthritis (RA) is a chronic inflammatory disease characterized by joint synovial inflammation, as well as cartilage and bone tissue destruction.当前处理RA策略可减少联合炎症,但处理选项仍代表稳定问题,因为它们不足并快速清除因此,数个药服系统(DDS)已经升级解决这一限制问题。可注入gellan口香糖(GG)水凝胶通过物理交叉法减少,并被建议为DDS,但这种交叉联通可产生水凝胶在生理条件中变弱倍感交叉连接度可调整 。 在本研究中,tyramine改制gellan口香糖并封装Bemethasone提高用RA治疗病人的特性和安全性物理化学结果显示,有可能用tyramine修改GG,代用率约30%显示高机械强度和抗药性 显示受控乙片松释放剖面Ty-GG流化凝胶也显示无细胞毒效,不会对代谢活动及原生细胞扩散产生消极影响此外,主要目标已经实现,因为证明比起单管贝美特松,Ty-GG流水胶效果更有效开发Ty-GG流格代表有希望DDS和可靠替代对RA.slink.spriet.com/article/10/10.s346/020855-9translRes.s.s/doi.org/10.1007s13346-020859 

Polymeric oral thin films (OTFs) were prepared by the casting method, combining gellan gum (GG), a water-soluble polysaccharide, and glycerol (Gly) as a plasticizing agent.GG-Gly电影被调查为使用fluconazole(生物药理分类系统第一类)作为示范药提供bucal药的潜在系统gly
Der Beitrag Polymeric oral thin films (OTFs) were prepared by the casting method, combining gellan gum (GG), a water-soluble polysaccharide, and glycerol (Gly) as a plasticizing agent.GG-Gly电影被调查为使用fluconazole(生物药理分类系统第一类)作为示范药提供bucal药的潜在系统gly低浓度下药时发生,而gly高浓度下药时则观察到mcoadhisive和机械性能显著退化。 解决所有这些问题的可能方法可能是添加hisypryl-spanids最大高度:无显示器内插字型风格:正常字体重量:正常平凡度量字型尺寸:12px文字缩进:0px文本对齐:左转文本转换式:无字母间距:正常字间间隔:正常溢出- wrap:常态白空格:即时拉普浮点数:无方向:i最大宽度:无0-px0px边界:0px0px边距:0pxposition: relative;" tabindex="0" role="presentation" data-mathml="">β-cyclodextrin (HP- Experimental results, supported by theoretical modeling, confirmed that gellan gum-low glycerol thin films including HP- Download the full article here or read it here Article Information: Adrover, A.!迪穆齐奥 LTrilli J白兰地CPalicelli P派特拉利托CasadeiM.Agelopyl-e-Cyclodextrin综合处理Gellan Gum Thin电影增强加载效率和 Mucoadhe


Methyl ester derivatives of alginic acid have been evaluated as potential multifunctional excipients for pharmaceutical direct compression.alginic酸作为平板配方前台使用有限,因为有某些缺陷,如低平板硬度和低压缩性这项工作的目标是通过下列方法提高这些属性 : [.]
>Der Betrag Methyl ester derivatives of alginic acid have been evaluated as potential multifunctional excipients for pharmaceutical direct compression.alginic酸作为平板配方前台使用有限,因为有某些缺陷,如低平板硬度和低压缩性这项工作的目标是提高这些特性,具体化通用于增强表单前接收器功能的化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工化工工工工工工工工工工工化工工工化工化工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工工总体说来,提高甲基化程度产生拉伸强度更高和压缩性更好的片片改代数显示延长分解时间,与引进疏水性后原生alginica最后,用微晶素纤维素和晶素来编译变异变异分解和填充/绑定试剂的功能多功能性测试href="https/www.scient.com/science/pii/S188087171538目标ss'blank'rels'胶片编译凝胶使用量对臂膀、肩膀、大腿内部或腹部生成薄生物沉积薄膜药物溶入胶片编织器中,并嵌入薄膜皮肤中胶片可起外部水库作用或限制向皮肤提供药物从而控制释放药Various gelling agents are listed in the following table (based on "Film forming systems for topical and transdermal drug delivery") Polymers are the foundation of the FFS and a variety of polymers are available for the preparation of these systems.实现所期望的胶片属性,这些聚合物可单独使用或与其他胶片组聚聚合物 Der Beitrag Film forming systems for topical and transdermal drug delivery erschien zuerst auf Pharma Excipients.
Gelling agent Properties Gellan gum •Anionic polysaccharide
•Gel properties depend on the presence of alts
•Excellent biocompatibility and degrades into non toxic products Carbomer (Carbopol®) Grades – ETD 2020, 171, 910, 934, 934P, 940, 1342 NF, 1971P •Excellent gelling efficiency in low concentrations of 0.25% to 1.5%
•pH dependent gelling
•Forms clear gel in water Carboxymethyl cellulose •Effective in concentrations of 4 to 6%.
•Stable between pH 2 and 10
•Incompatible with ethanol HPMC (E series, F series, K4M, K15M, K100M) •Effective in concentrations of 2–6% as gelling agent
•Produces a smooth, clear spreadable gel Hydroxy ethyl cellulose •Effective in concentration of 0.5–1%
•Has thickening and stabilizing properties Poloxamer (polyethylene–polypropylene glycol co-polymer) Grades – 124, 182, 188, 407 •Used in the concentrations of 20–30 %
•Thermoreversible gelation-liquid at refrigerated conditions and gel at room temperature Sepineo P 600
(acrylamide/sodium acryloyldimethyltaurate copolymer)•Concentration of 0.5–5% used as gelling agent
•It has thickening properties in aqueous or organic media over a wide pH range Film Forming Polymers
Polymer Properties Hydroxypropyl
Methylcellulose (HPMC)
HPMC (E4M, E15, E50M K4M)• Produce a light, non-greasy uniform film with good texture
• Do not interact significantly with other ingredients
• Surface active agent, therefore adsorbs water providing easy dispersion, lubricity and comfort feel in occlusive state on application to skin Ethyl cellulose (EC) • Nontoxic, nonirritating, nonallergic material
• Good film forming properties that form tougher films Hydroxypropyl cellulose • Nonionic, pH insensitive polymer
• Water soluble Polyvinyl pyrrolidine (PVP)(PVP K30, PVP VA64 • Solubility in water and other solvents
• Adhesive and binding property
• Acts as a bioavailability enhancer Polyvinyl alcohol (PVA) • Water soluble
• Excellent film forming and adhesive properties
• Nontoxic and biocompatible Chitosan • Excellent film forming ability
• Opens the tight junctions of mucosal membrane, thereby enhancing the paracellular permeability and penetration of drug
• Controls drug release Eudragit (polymethacrylates copolymer)
Eudragit RS 100, RL 100, NE, RS 30D, S 100• Transparent, elastic, self-adhesive
• Good adhesion to the skin Silicones
Polydimethylsiloxane (PDMS)• Water vapor permeable film
• Adequate substantivity and durable film Acrylates copolymer
Avalure® AC 118, AC 120• Tough, breathable, abrasion resistant films

The objective of the present study was to develop a pH-sensitive drug delivery system by using polymethylmethacrylate-grafted gellan gum (PMMA-g-GG).PMMA-g-GG使用红牛启动机硝酸铵合成自由基聚合响应并编成数组同聚物grafting参数等 [.]
Der Betrag

The objective of the present study was to develop a pH-sensitive drug delivery system by using polymethylmethacrylate-grafted gellan gum (PMMA-g-GG).PMMA-g-GG使用红牛启动机硝酸铵合成自由基聚合响应并编成数组同聚物剖分参数,例如百分比和效率计算,单数和启动者集中效果研究分包产值。
优化用最优响应面法完成批量配接率更高并特征为元素分析(CHN)、FT-IR、DSC、PXRD、sup>1 H-NMR和SEM此外,急性口服毒性研究及组织病理分析显示系合口香糖的非毒性和生物兼容性Mitformin盐粒子使用MMA-GG详细描述并评估生物兼容性和有效性PMMA-g-GG配方M4展示PH敏感度和持续释放12H和发布剖面活性效率研究显示预制配方有很有希望的抗电算潜力PMA-gG-GG配方在不久的将来可归结为辅助抗糖尿病新药提供系统。
s/link.sprianger.com/artems/10/10.s346/020-00777translRes. (2020).https://doi.org/1017s13346-020077 Der Beitrag
It is a challenge to safely administer sustained release medicines to patients with dysphagia.持续释放片片片不可压碎,多片段粒度大可降低患者可接受性研究的目的是开发即时推送器,为有缺陷的病人持续释放微粒干粉 [.]
Der Betrag

It is a challenge to safely administer sustained release medicines to patients with dysphagia.持续释放片片片不可压碎,多片段粒度大可降低患者可接受性研究的目的是开发即时推送器,为有缺陷的病人持续释放微粒含凝胶物剂的干粉混合体,如悬浮钠和钙离子水分20毫升水分组成10分钟果冻纹理即时Jellies展示出可与商业网际JELLIES相匹配性能,外观性能、风学性能/文体性能和体外吞咽性能人造喉模gliclazide持续释放微粒s持续滑动释放超过15小时,微粒注入jellies显著降低药物释放率新药提供系统为向有缺陷的病人持续放药的长期挑战提供以病人为中心的解决办法,并有可能用于儿科病人 下载全出版物:https://www.pharmaexbens.com/wp-content/uploads/2020/05/Easy-to-Swarow-Instant-Formations-slift-Release-Gliclazide-Delivery.pdfMcAuley, Fang Liu
Publication: Journal of Pharmaceutical Sciences
Publisher: Elsevier https://doi.org/10.1016/j.xphs.2020.04.018
Keywords: Dysphagia, Paediatric, Geriatric, Controlled release, Swallowing, Microparticles, Cellets 100, Protanol GP 1740, Kelcogel F, HPMC, Methocel E5, Sentry Polyox WSR N10 LEO NF, Mini-Glatt, fluidized bed
Der Beitrag Easy to Swallow "Instant" Jelly Formulations for Sustained Release Gliclazide Delivery erschien zuerst auf Pharma Excipients.

The aim of the present study was the development of a novel gellan gum derivative exhibiting mucoadhesive properties for nasal application.实现这一点后,Minoino群落被引入聚合骨架所生成合成产品特征化为附氨基组数,水分化、Zeta潜力和凝胶特征mcoadheisness unity
DerBeitrag The aim of the present study was the development of a novel gellan gum derivative exhibiting mucoadhesive properties for nasal application. Accomplishing this, amino groups have been introduced to the polymeric backbone.所生成合成产品特征化为附氨基组数,水分化、Zeta潜力和凝胶特征以旋转圆柱和抗拉研究对 Mucoadhisive研究协同性进行评估紧接红细胞/细胞毒性评价,调查对鼻膜上下角细胞周期的影响。 Results显示联通率达1259.50+75.98mol/g聚合物并加速水化衍生物聚合物/粘合物粘合度增加32倍和14倍扩展粘合时间验证了未变凝胶增强粘合度耐用性研究显示共加工作9倍和3.75倍最大分遣队细胞膜没有严重缺陷CBF研究证明应用新衍生物可逆抑制性 。 aahrefs/www.scient.com/science/abs/bs/s014113020299gems='blank'rels


Gellan gum was chemically modified by the reaction with methacrylic anhydride to produce derivatives with 6, 14 and 49% methacrylation.iNMR和FTIR分光镜确认这些衍生物的结构和替代度与原状凝胶相比,这些衍生物多防水并形成水中的扰动解析法gellan-gum-forms-of-pilapine/

Gellan gum was chemically modified by the reaction with methacrylic anhydride to produce derivatives with 6, 14 and 49% methacrylation.iNMR和FTIR分光镜确认这些衍生物的结构和替代度与原状凝胶相比,这些衍生物多防水并形成水中的扰动解析法体外研究配方含凝胶素及其甲状腺衍生物显示,甲状腺变异会增加保留物上。
/p>ivore实验含凝胶和甲状腺变衍生物pilcarpine盐化物配方证明所有聚合物都显著增强药效,但观察聚氨酸酯优性能为6%Download the full publication hereThis video abstract describes our paper "Gellan gum and its methacrylated derivatives as in situ gelling mucoadhesive formulations of pilocarpine: in vitro and in vivo studies" published in International Journal of Pharmaceutics https://authors.elsevier.com/a/1aW0-1…
Der Beitrag Gellan gum and its methacrylated derivatives as in situ gelling mucoadhesive formulations of pilocarpine: In vitro and in vivo studies erschien zuerst auf Pharma Excipients.