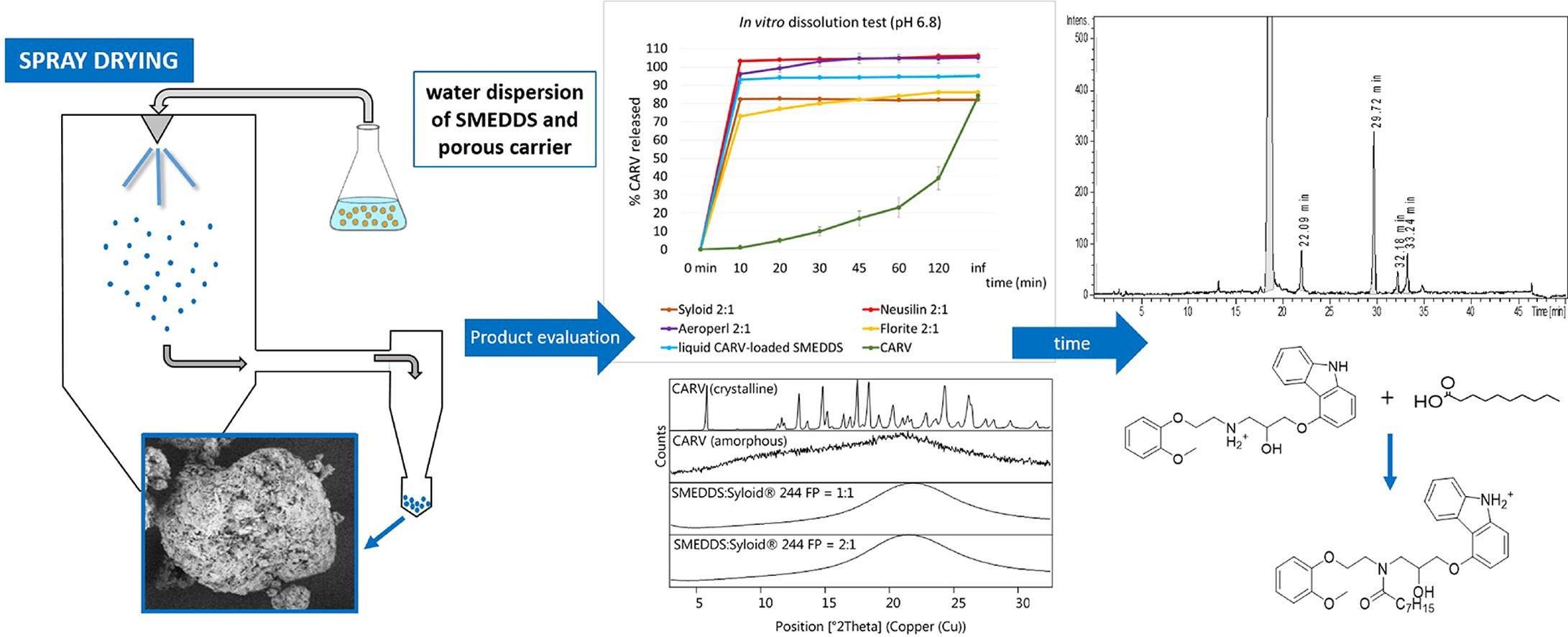
In this study, various formulations of solidified carvedilol-loaded SMEDDS with high SMEDDS loading (up to 67% w/w) were produced with the spray drying process using various porous silica-based carriers.smedds/''s评估喷雾干燥法生成的固态刻装SDDS
Deberag
In this study, various formulations of solidified carvedilol-loaded SMEDDS with high SMEDDS loading (up to 67% w/w) were produced with the spray drying process using various porous silica-based carriers.The process yield was improved with higher atomization gas flow rate during the spray drying process and with prolonged mixing time of dispersion of liquid SMEDDS and solid porous carriers prior to the spray drying process.
Highlights
Spray drying enables production of s-SMEDDS with high SMEDDS loading (up to 67% w/w).
Mixing time of dispersion before spray drying has impact on process yield.
Carrier type has impact on process yield, drug content and s-SMEDDS characteristics.
Carvedilol forms amides with fatty acids from the oily components of SMEDDS.
Depending on the choice of the carrier and the SMEDDS:carrier ratio in solid SMEDDS, different drug loading, self-microemulsifying properties, drug release rates, and released drug fractions were obtained.产品展示快药释放,原因是保存自微分解特性和缺晶形刻度,XRD和Raman映射证实了这一点。
A稳定化研究期间减少药量得到观察和调查这部分归结于该药的化学降解LC-MS方法测定的关键降解产物由capilol现场反射SMEDDS中的脂肪酸组成。
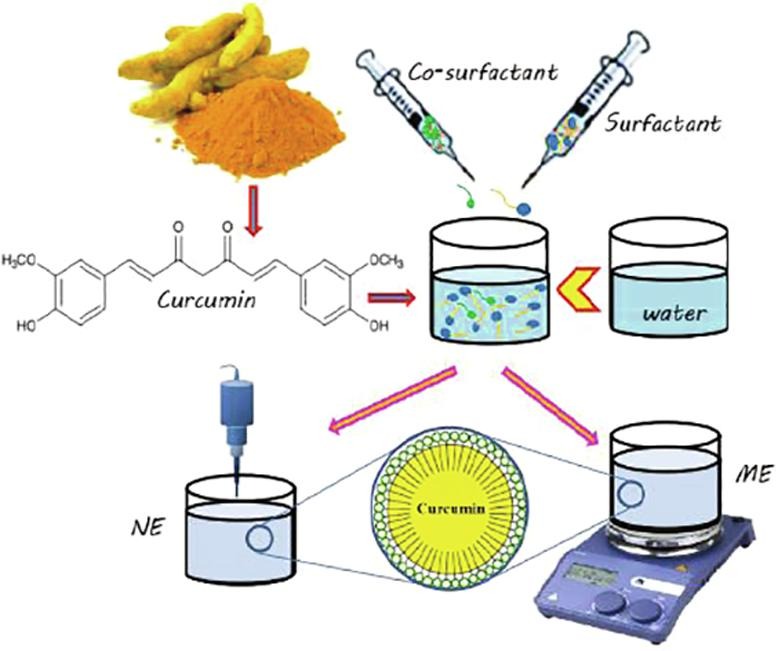
Phenolic compounds (phenolics) have received great attention in the food, pharmaceutical and nutraceutical industries due to their health-promoting attributes.但它们的广泛使用有限,主要是因为它们在处理条件和/或肠胃交互作用下差分水和不稳定性,影响生物可用性/生物可用性hrefss/www.pharmaexsubjects.com/news/encapsation-phenclips-comps-2

Phenolic compounds (phenolics) have received great attention in the food, pharmaceutical and nutraceutical industries due to their health-promoting attributes.However, their extensive use is limited mainly due to their poor water dispersibility and instability under both processing conditions and/or gastrointestinal interactions, affecting their bioavailability/bioaccessibility.
Highlights
Phenolics have received great attention in the food, pharmaceutical and nutraceutical industries.
Their extensive use is limited mainly due to their poor water dispersibility and instability.
Nano/microemulsions have been widely used to encapsulate phenolics.
Stability, encapsulation efficiency, cytotoxicity, bioavailability and releasing rate are discussed.
Therefore, different nanocarriers have been widely used to encapsulate phenolics and overcome the aforementioned challenges.据我们所知,除多项研究外,迄今尚未发布关于微模和纳米模子封装的全面审查本研究因此试图审查Penlips加入MEs和NES.
并讨论开发系统的基本特征,如稳定性、封装效率、细胞毒性、生物可用率和释放率MEs和NEs都证明是封装保护Penlices的适当工具。Der Beitrag Encapsulation of phenolic compounds within nano/microemulsion systems: A review erschien zuerst auf Pharma Excipients.
Dry eye disease (DED) or keratoconjunctivitis sicca is a chronic multifactorial disorder of the ocular surface caused by tear film dysfunction.症状包括干燥性、刺激性、不适和视觉干扰,标准处理包括润滑油和局部类固醇使用二次发炎在开发传播这种虚弱条件中起着突出作用[.]
Der Beitrag
Dry eye disease (DED) or keratoconjunctivitis sicca is a chronic multifactorial disorder of the ocular surface caused by tear film dysfunction.症状包括干燥性、刺激性、不适和视觉干扰,标准处理包括润滑油和局部类固醇使用二次炎症在开发传播这一虚弱条件中起着突出作用 。
解决此点,我们已经调查创新药物提供系统实验规模开发使用 dexethasone/emoji/13.0.172/2122.png' altss/www.pharmaexccidents.com/product/labrafac-lipoptile-wl-1349/最大高度:1em;"/ > 亲脂载量/a快速筛选实验室规模预编程后,所选配方以实验规模编译,粒度为19.51+++0.5Nm,封装效率99.6+++0.5%,PDI0.08和4°C扩展稳定性6个月观察发现,这种潜在的眼科配方具有高可耐性并内化能力,在ex vivoporcine角膜研究中展示相似行为,建议奥氏表面适当分布。
p>serve,ELISA用于研究实验级配方对各种炎症生物标志的影响最成功的解甲松装入NLC显示单方三叉松TNF-A生产5倍下降,MMP-9和IL-6相仿结果易编译性、可扩缩性、性能和生物标志分析显示,NLC配方可成为DED专题处理的可行选项。
ahref='https//www.pharmaexbiders.com/wp-content/uploadss/2021/06/Dexicathone-Loaded-Nanostrationd-Lipid-Carriers-s-the-Treatment-Dry-Eye-Disaces.pdf'丹达穆迪MRani S.贝海盖尔E贝哈尔G肯特D欧里利NJ欧多诺万市麦克劳林市菲茨亨里市deximetasoni-loadnationlipid载体处理干眼疾病。 药理 2021 , 13 905https://doi.org/10.3390/pharmaceutics13060905Der Beitrag Dexamethasone-Loaded Nanostructured Lipid Carriers for the Treatment of Dry Eye Disease erschien zuerst auf Pharma Excipients.
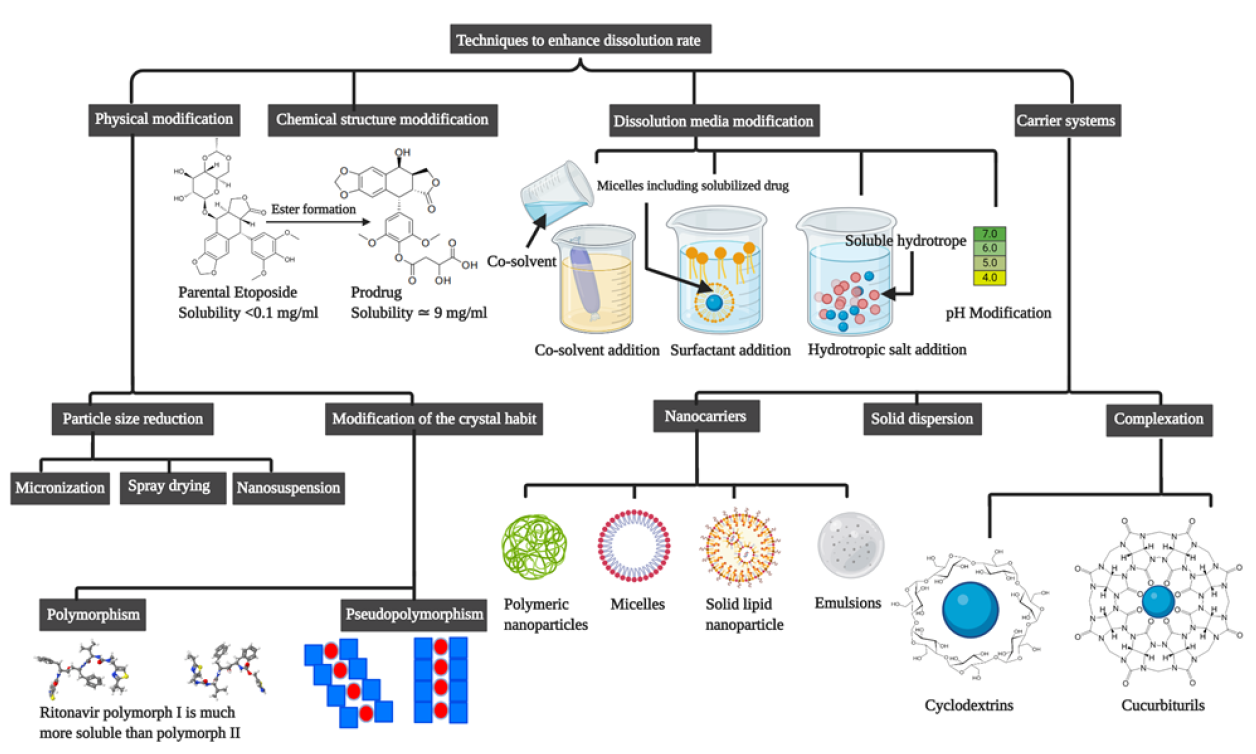
Solid dispersion (SD) is one of the oldest and widely utilized techniques to improve the solubility of slowly dissolving drugs.使用各种新兴技术制作自定义多方法设计准备自定义方法,例如kneading、联运、聚变、溶剂蒸发和各种溶剂相关方法eschienzuftahrefss/www.pharmaexsubjects.com/news/soli-disclipation-rate/

Solid dispersion (SD) is one of the oldest and widely utilized techniques to improve the solubility of slowly dissolving drugs.使用各种新兴技术制作自定义多方法设计准备自定义,方法有kneading、联运、聚变、溶剂蒸发和各种溶剂相关方法。
最近多次努力提高应用方法的可扩展性,结果为新编程方法,如KinetiSol,
See the new webinar about HME and ASD here:
Der Beitrag Solid Dispersion as a Technical Solution to Boost the Dissolution Rate and Bioavailability of Poorly Water-Soluble Drugs erschien zuerst auf Pharma Excipients.

In case of colon-specific delivery of therapeutic agents through oral route, microbial/enzyme-triggered release approach has several advantages over other approaches due to unique microbial ecosystem in the colon.多单元运算器比单单元运算器有边际性在探索的不同材料/聚合物中,外壳似乎是一种有前途的生物聚合物,用于制造微生物触发式结核载体Pectin
Beitrag In case of colon-specific delivery of therapeutic agents through oral route, microbial/enzyme-triggered release approach has several advantages over other approaches due to unique microbial ecosystem in the colon.多单元运算器比单单元运算器有边际性在探索的不同材料/聚合物中,外壳似乎是一种有前途的生物聚合物,用于制造微生物触发式结核载体外壳通过共生酶具体降解,但上胃寄生酶无法理解。 
国际药理学杂志,2021年https://doi.org/10.1016/j.ijpharm.2021.120814.
Der Beitrag Pectin based multi-particulate carriers for colon-specific delivery of therapeutic agents erschien zuerst auf Pharma Excipients.
Nanomedicines based on poly(lactic-co-glycolic acid) (PLGA) carriers offer tremendous opportunities for biomedical research.PLGA基础数系统已经获得FDA和欧洲药管局批准并广泛用于诊所治疗或诊断疾病,但目前没有PLGA纳米医学配方

Nanomedicines based on poly(lactic-co-glycolic acid) (PLGA) carriers offer tremendous opportunities for biomedical research.Although several PLGA-based systems have already been approved by both the Food and Drug Administration (FDA) and the European Medicine Agency (EMA), and are widely used in the clinics for the treatment or diagnosis of diseases, no PLGA nanomedicine formulation is currently available on the global market.
One of the most impeding barriers is the development of a manufacturing technique that allows for the transfer of nanomedicine production from the laboratory to an industrial scale with proper characterization and quality control methods.
This review provides a comprehensive overview of the technologies currently available for the manufacturing and analysis of polymeric nanomedicines based on PLGA nanoparticles, the scale-up challenges that hinder their industrial applicability, and the issues associated with their successful translation into clinical practice.
Download the full article as a PDF here or read the article here
Introduction
Nanotechnology is among the most promising Key Enabling Technologies (KETs) that can provide innovative and radical solutions to the unmet needs of society (Soares et al., 2018, Tinkle et al., 2014).今天,纳米技术触及人生活的方方面面,包括医学,并产生最重要的医学健康研究新领域之一:纳米医学(欧洲委员会,2020年)。纳米医学应用纳米材料和纳米技术应对诊断、监测、控制、预防和治疗疾病方面的挑战(Agrahari和Hiremath,2017年,Patra等,2018年,Soares等,2018年,Tinkle等,2014年)。纳米医学配制成药物提供系统通常涉及活性药料,这些药料或封装或混入纳米载体矩阵(Murthy,2007年)。载体材料可基于无机性(例如,金属纳米粒子、半导体量子大小和形状(Biju等人,2008年;Tagit等人,2015年;Tagit等人,2017年;Tagit等人,2011年)或有机纳米结构,包括(并不限于)聚合物(Kumari等人,2010年)、drimerers(Giles和Frechet,2005年)、小鼠(Katoka等人,2012年)、亲脂类(Aravi等人,20177年)、固脂纳米粒子(Mukherjee等人,2009年)和聚合物作用药构件(API)并发(Larson和Ghandehari,2012年)。纳米医学在纳米尺度上精细设计以引入各种惠益,如保护治疗剂不退化、提高溶解度和生物可用性、改善药代物学、降低毒性、提高治疗效果、降低API免疫性、定向交付和单系统同时诊断和处理选项(Agrahari和Hiremath,2017年,Patra等2018年)。传输器应 三) 在血包中为“stealthy”,以有效避免复元系统筛选,但 iv) 接触并渗透目标动作网站右细胞(Sun等,2012年)。纳米载体多词性能为设计与性能提供了巨大契机,这要归功于PLGA使它成为理想纳米载体的各种特性(Han等人,2016年;Makadia和Siegel,2011年;Singh等,2014年)。PLGA是一种生物可降解和生物兼容聚合物,有多种降解时间,可按分子权重和共聚比调适PLGA可溶解常见溶剂,包括乙酮、氯化溶剂和乙乙酸乙酯,可处理成几乎各种形状和大小,并可封装几乎各种大小的分子(Gentile等人,2014年;Makadia和Siegel,2011年;Södergord和Stolt,2002年)。PLGA聚合物大都测试成药箱、蛋白质和各种其他宏模类,如脱氧核糖核酸、RNA和pitides(Jain,2000年;Makadia和Siegel,2011年)。除聚合物化学成分和分子权外,PLGA纳米载体物理属性,如大小、形状、表面积对容量比等,可进行“调整”,以获取理想发布剖面图(Gentile等,2014年;Makdia和Siegel,2011年)。
This study provides an overview of the technologies available for the production and analysis of PLGA nanoparticles, the scale-up challenges that hamper their industrial applicability, and the issues associated with their successful translation into clinic.
Article information: Maria Camilla Operti, Alexander Bernhardt, Silko Grimm, Andrea Engel, Carl Gustav Figdor, Oya Tagit.spanspan样式=text- transform:初始化;spanscriptroductions制造技术概述与挑战https://doi.org/10.1016/j.ijpharm.2021.120807.
Der Beitrag PLGA-based nanomedicines manufacturing: Technologies overview and challenges in industrial scale-up erschien zuerst auf Pharma Excipients.
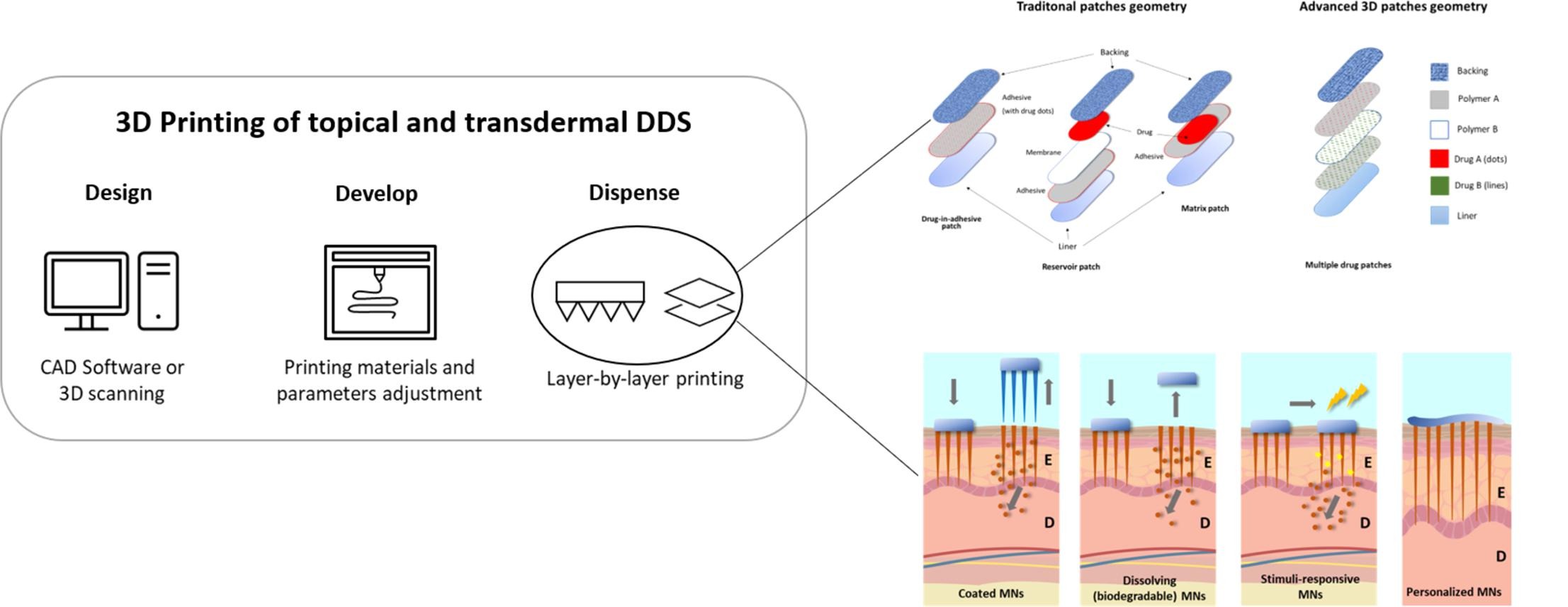
The incorporation of 3D printing technologies in the pharmaceutical industry can revolutionize its R&D, by providing a simple and rapid method to produce tailored one-off batches, each with customized dosages, different compounds, shapes, sizes, and adjusted release rates.具体地说,这类技术可有利于开发热点和跨面药提供系统,包括 [.]
Der Beitrag The incorporation of 3D printing technologies in the pharmaceutical industry can revolutionize its R&D, by providing a simple and rapid method to produce tailored one-off batches, each with customized dosages, different compounds, shapes, sizes, and adjusted release rates.具体地说,这类技术可有利于开发专题和跨式药物提供系统,包括补丁和微软码片。 
最近数项研究被选择支持本文所探讨主题的相关性并讨论这些打印技术的局限性,包括规范、质量和安全问题。
The objectives of this investigation were to study the evolution in blend state of adhesive mixtures containing the active pharmaceutical ingredients (APIs) salbutamol, budesonide and AZD5423 and to study the relationship between blend state and dispersibility of the mixtures, as assessed by the fine particle fraction (FPF).各种罚款串合相容性 [.]
derBeitrag

The objectives of this investigation were to study the evolution in blend state of adhesive mixtures containing the active pharmaceutical ingredients (APIs) salbutamol, budesonide and AZD5423 and to study the relationship between blend state and dispersibility of the mixtures, as assessed by the fine particle fraction (FPF).
A series of adhesive mixtures of varied fines concentration were prepared for each API using the same type of carrier.基于视觉检验和火药力学,混合状态被识别并归纳为每个API混合状态地图使用装有ScreenHaler快速筛选撞击器研究混合物的偏差性混合状态演化因混合状态宽度而异粘合层结构也因API而异,从相对统一层到小聚聚散于载波表面的多维层不等三大API表示FPF和罚款集中度之间相似的混合关系。
所有API粘合组合结构进化三大状态,FPF-Fines集中剖面总体形状可解释混合状态演化adsion层结构是一个重要因素解释混合状态-dhiv-mixtices.pdf目标='blank'rel='noopener'混合状态与含有活性药素的粘合混合物分布关系问题,国际药理学杂志:X卷32021https://doi.org/10.1016/j.jpx2020100069.
Der Beitrag
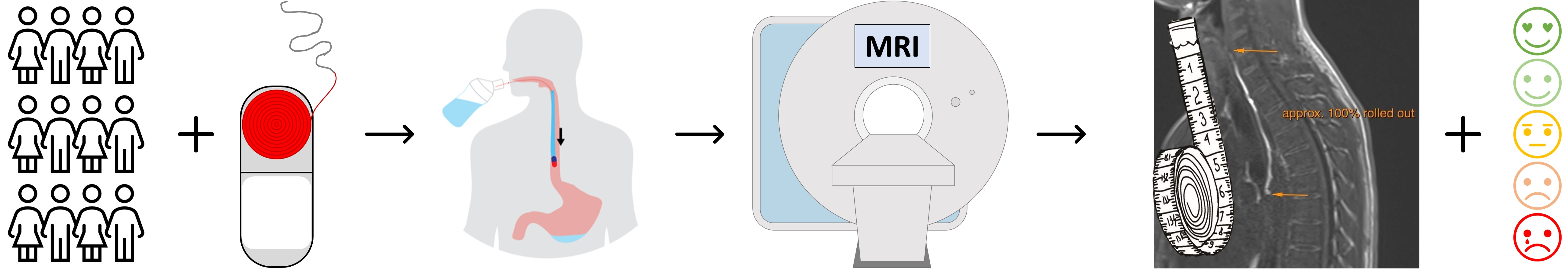
There are no methods for specific local application of active substances to the mucosa of the esophagus to treat eosinophilic esophagitis or other esophageal diseases.并接受12名健康志愿者新模块化药提供概念EsoCap系统EsoCap [.]
Der Beitrag There are no methods for specific local application of active substances to the mucosa of the esophagus to treat eosinophilic esophagitis or other esophageal diseases.并接受12名健康志愿者新模块化药提供概念EsoCap系统EsoCap系统首次允许定点投放聚合胶片的食道粘膜上。 接受度通过标准问卷确定,通过MRI扫描生成设备并实现功能EsoCap系统两个不同的搭建测试:一个高密度为0.4g/cmsup3 ,另一个高密度为1.0g/cmsup3 functionality-and-aceptation-of-EssoCap-System-A-Nevled-Drug-Delipsy-Technology-a-硬凝胶囊长23.3毫米直径8.2毫米从Wepa购买polyvinyl酒精(PVA 18-88)由s/www.pharmaexccidents.com/merck-a-leader-in-science/glycerol制作胶片所需的增塑剂是从Caelo获取的(德国希顿市)。淡化水用作溶剂Formutura(荷兰Nijmegen)食品级多级酸丝化3D打印酒杯与应用器前曾描述过 [squal='html-br'标题='''hrefss/www.mdpi.com/1999-4923/13/628/htm#B8-pharmecutics-13-00828'>8 药理学 2021 , 13 828https://doi.org/10.3390/pharmatutics1306028 
Leucine is a promising excipient with several applications in the development of inhalable spray-dried powder of high- and low-dose drugs.添加 Leucne对所产生粒子的喷雾化和物理稳定性产生显著增强效果高亮性能Leucine物理特性和喷雾干燥行为设计处理参数控制lecine功能[.]
Der Beitrag

Leucine is a promising excipient with several applications in the development of inhalable spray-dried powder of high- and low-dose drugs.lecine加法对所产生粒子的浮质化和物理稳定性产生显著增强效果。
lblockte>此外,我们强调目前Leucene监管状态深入了解对未来更有效地利用lecine非常重要,无论是干粉吸入器配方还是其他药剂应用都如此。More on leucine
Article information: Nasser Alhajj, Niall J.欧里利 海伦卡斯卡特Leucine原创者喷干粉吸入,《药发现今日》2021年https://doi.org/10.1016/j.drudis.2021.04.009.
Der Beitrag Leucine as an excipient in spray dried powder for inhalation erschien zuerst auf Pharma Excipients.
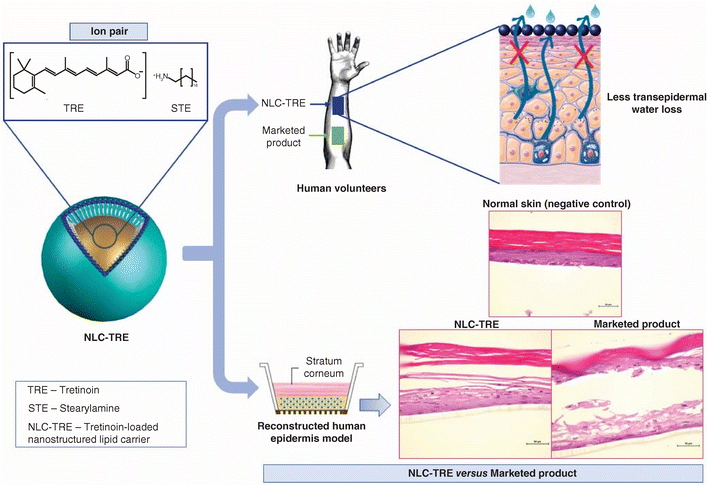
DerBeitrag Aim: To enhance the tretinoin (TRE) safety profile through the encapsulation in nanostructured lipid carriers (NLC). Materials & methods: NLC-TRE was developed using a 23 experimental factorial design, characterized (HPLC, dynamic light scattering, differential scanning calorimetry, x-ray diffraction analysis, transmission electron microscopy, cryo-transmission electron microscopy) and evaluated by in vitro studies and in healthy volunteers. Results: The NLC-TRE presented spherical structures, average particle size of 130 nm, zeta potential of 24 mV and encapsulation efficiency of 98%.NLC-TRE保护TRE避免氧化(p <0.0001)并推广上下文定位(p <0.0001)与营销产品相比,两者均0.05%TREThe in vitro assay on reconstructed human epidermis and the measurement of transepidermal water loss in healthy volunteers demonstrated an enhanced safety profile in comparison to the marketed product (p < 0.0002). Conclusion: The NLC-TRE enhances the epidermal targeting and safety profile of TRE, representing a potential safer alternative for the topical treatment of skin disorders using TRE. Article information: Flávia A Lima, Raquel VR Vilela, Rodrigo L Oréfice, Izabela R Silva, Eduardo CO Reis, Larissa AC Carvalho, Silvya S Maria-Engler, Lucas AM Ferreira, and Gisele AC Goulart.span类sspan/span/span/span类smessions/spans/span
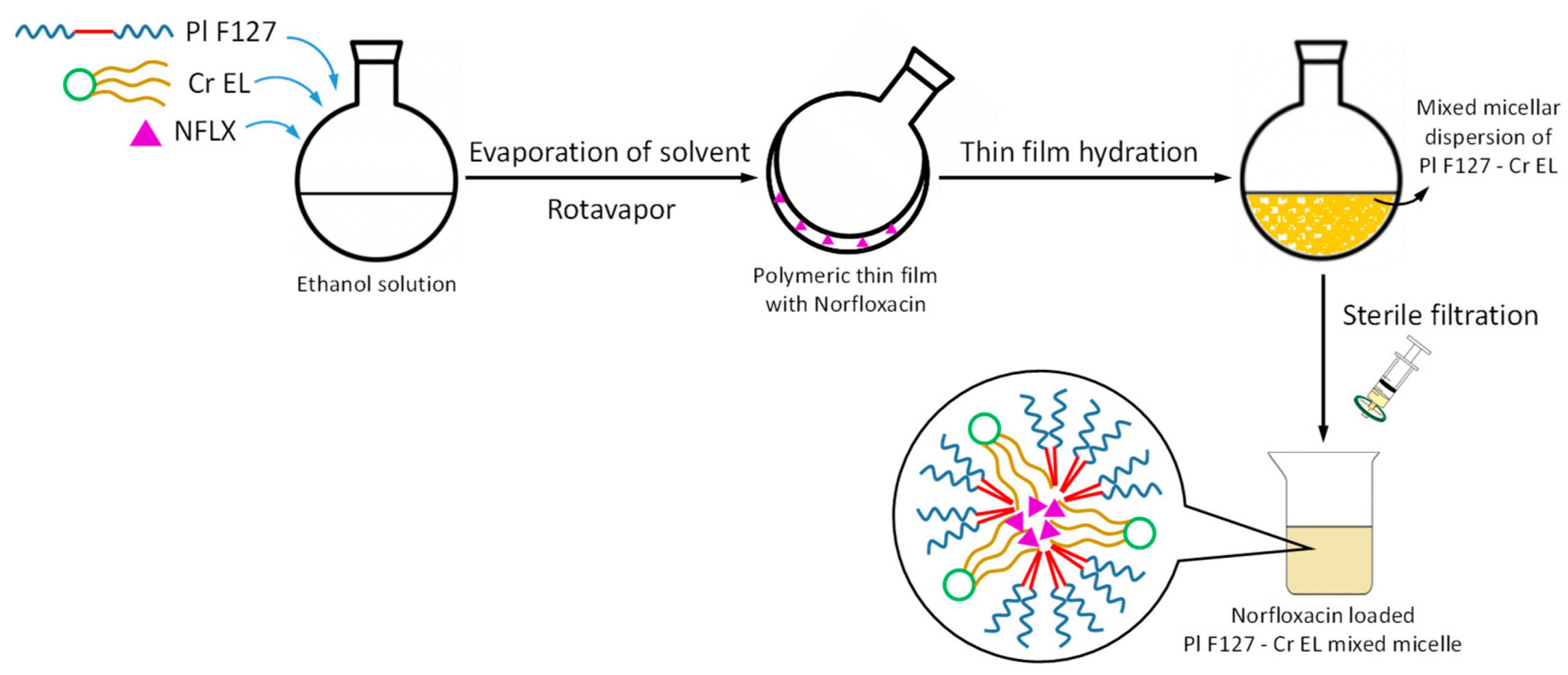
In this work, novel polymeric mixed micelles from Pluronic F127 and Cremophor EL were investigated as drug delivery systems for Norfloxacin as model antibiotic drug.确定表面活性剂最优模比,以降低临界小鼠富集度并让载体最小表面活性富集粒子大小、zeta潜力和封装 [.]
DerBetrag

In this work, novel polymeric mixed micelles from Pluronic F127 and Cremophor EL were investigated as drug delivery systems for Norfloxacin as model antibiotic drug.确定表面活性剂最优模比,以降低临界小鼠富集度并让载体准备最小表面活性富集度。
小鼠体外释放Norfloxa混合小鼠对正常纤维化MRC-5细胞表现出良好的生物兼容性,同时在所有高浓度小鼠系统都发现某些细胞毒性载药混合小鼠配方比CLSI推荐标准菌株(span类='html-itics'/span>ATCC 25923,span类='html-itaclis'/span>ATCC 25922.span类='html-italis'aruginosa 5399临床菌株显示在所有测试小鼠系统对Norfloxacin敏感度低检测结果显示,Cremophor EL-Pluronic F127混合小鼠可视之为强水杀菌药新式释放系统。
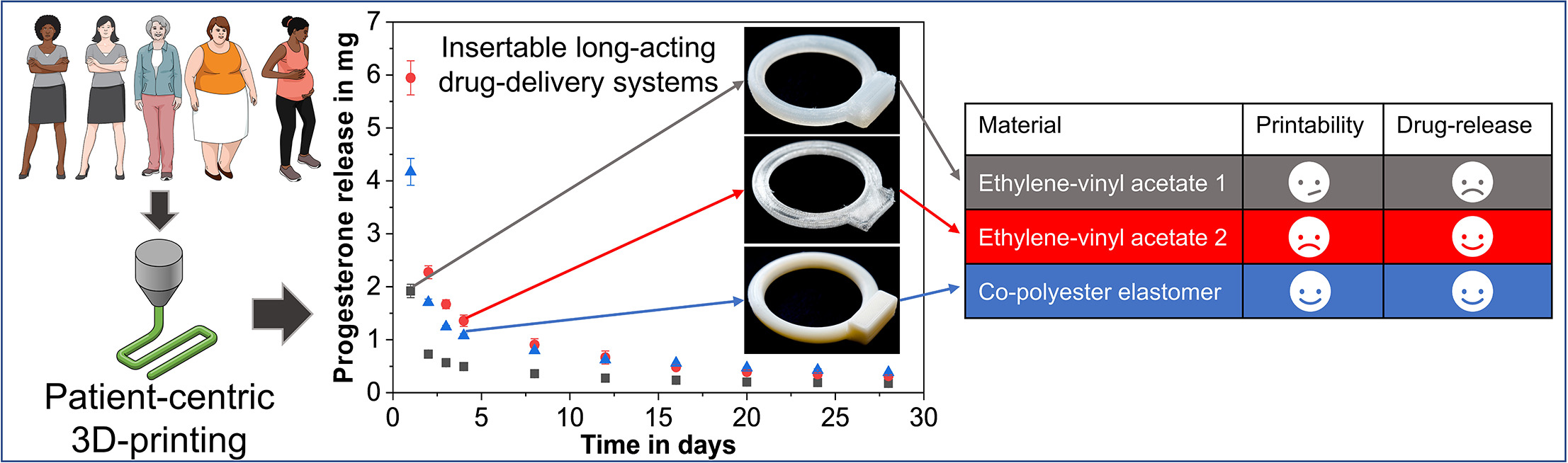
To improve patient compliance and personalised drug delivery, long-acting drug delivery devices (LADDDs), such as implants and inserts, greatly benefit from a customisation in their shape through the emerging 3D-printing technology, since their production usually follows a one-size-fits-most approach.三维打印LADDs使用受限制,但主要受三维可打印应用不足的限制 [.]
Der Beitrag To improve patient compliance and personalised drug delivery, long-acting drug delivery devices (LADDDs), such as implants and inserts, greatly benefit from a customisation in their shape through the emerging 3D-printing technology, since their production usually follows a one-size-fits-most approach.The use of 3D-printing for LADDDs, however, is mainly limited by the shortage of flawlessly 3D-printable, yet biocompatible materials. A novel 3D-printing excipient for long-acting drug delivery devices is introduced. A mechanistic understanding of material requirements for 3D-printing is presented. A novel progesterone immersion strategy for 3D-printed devices is developed. The novel excipient shows comparable drug permeability to ethylene-vinyl acetate. The present study tackles this issue by introducing a novel, non-biodegradable material, namely a polyester-based thermoplastic elastomer (TPC) – a multi-block copolymer containing alternating semi-crystalline polybutylene terephthalate hard segments and poly-ether-terephthalate amorphous soft segments.下一步详细描述材料3D可打印性,通过机械、风学和热学分析发现优于传统聚合物(ene-evenyacates),本研究首次建立对progeTPC展示了广选移植尺寸和复杂地理特征的3D可打印性。 
Highlights
Der Beitrag Novel polyester-based thermoplastic elastomers for 3D-printed long-acting drug delivery applications erschien zuerst auf Pharma Excipients.
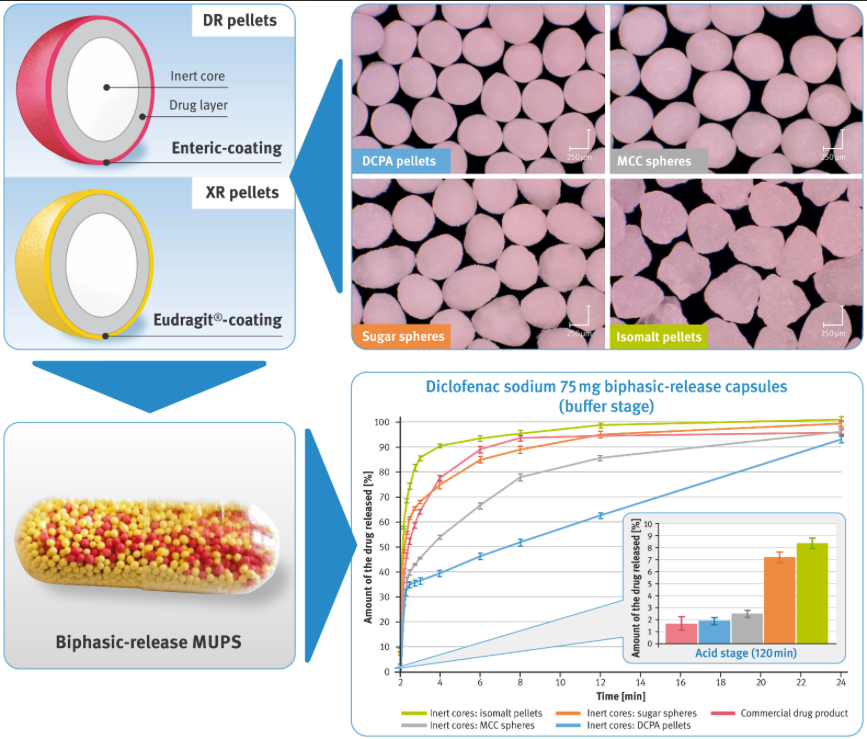
Novel calcium phosphate-based starter pellets were used to develop a biphasic-release multiple-unit pellet system (MUPS) with diclofenac sodium as a model drug in the form of hard gelatin capsules.编译对应配方供比较使用,这些配方基于微晶素细胞素、苏格鲁斯和异默特制成的惰性核心开发系统由两类组成 [.]
DerBeitrag Novel calcium phosphate-based starter pellets were used to develop a biphasic-release multiple-unit pellet system (MUPS) with diclofenac sodium as a model drug in the form of hard gelatin capsules.编译对应配方供比较使用,这些配方基于微晶素细胞素、苏格鲁斯和异默特制成的惰性核心开发系统由两类药层粒子组成,实现不同的释放模式:延迟释放(内装)和扩展释放分解特征使用折合法和生物相关法检验,这些方法反映进食条件和禁食条件。整理结果时使用等效商业产品,但直接分解技术编译弗朗基维茨M赫斯T卡尔K加达M达布劳斯卡J库比亚克市帕斯科夫斯卡维特M高手D加巴茨G哈兹纳尔-加尔巴茨s/www.mdpi.com/1999-4923/13/6805#cite目标s药理2021 13 805https://doi.org/10.3390/pharmacetical130605 
Diclofenac钠(印度孟买Amoli组织)。a hrefs/pherations-cm/mWernerGmbH,Torensch,德国)bellet启动器bellessqahrefss/www.pharmaexbens.com/product/galeniq-960Film coating systems: Vivacoat® FM-1M 000 (JRS Pharma, Rosenberg, Germany), Aquarius® Control ENA (Ashland, Covington, KY, USA), Eudragit® RL30D and Eudragit® RS30D (Evonik, Darmstadt, Germany).透明硬胶囊壳,大小为00(Pharmapol ArzneimitelveribGmbH,Dägeling,Darzneittel75 mg/cloDuo一系列性能测试显示,控制药用从开发配方分解显然与惰性核水溶性相关观察发现水分启动器粒子(MCC球体和DCPA粒子)比可溶甘球或Isomat粒子能更好控制释放率在后一种情况下,可以假设使用效率更高的聚合物或更大量聚合物将导致拆解率下降。MCC和DPA启动粒子为提高生产率和控制小量聚合物药解速率提供了极有吸引力的机会。磷酸盐粒子似极有希望。
Nasal route of administration offers a unique opportunity of brain targeted drug delivery via olfactory and trigeminal pathway, providing effective CNS concentrations at lower doses and lower risk for adverse reactions compared to systemic drug administration.最近有人提议它为glucocortipse控制病人神经发泡过程选址 [.]
Der Beitrag Nasal route of administration offers a unique opportunity of brain targeted drug delivery via olfactory and trigeminal pathway, providing effective CNS concentrations at lower doses and lower risk for adverse reactions compared to systemic drug administration.最近有人提议用它作为选择通道 控制重科维德19患者神经发火过程适当的送货系统提高效率,但尚需出现本文介绍开发可喷脑反三叉松磷酸钠粉发平台DSP加载微信箱最优使用逐项质量方法,与可溶性惰性载波混合(mannitol或lactose单水合物)。火药混合特征表现为同质性、流性、可喷雾性、体外生物兼容性、渗透性和粘合性Nasal沉降研究使用3D打印鼻腔模型Mannitol比Lactose提供更好的火药混合流属性微光与manitol混合保留或放大它们的粘合性能并增强DSP渗透性跨侧膜模型屏障DSP分量沉入嗅觉区达17.0%,暴露开发火药平台对定向嗅觉投送的潜在作用ahrefs/www.pharmaexbens.com/?attachment_id=225826乌格里那州ISpoljaricDAmidžicKlari雅各布希奇布拉拉PerkušićM佩皮奇洛夫里奇 J萨尔松V赛福季奇库库克扎德拉维克市Kalogjera L.哈夫纳Aa hrefss/www.mdpi.com/1999-4923/13/6/795/htm目标ssshttps://doi.org/10.3390/pharmacetics1306095 
细胞生物兼容性和渗透性试管研究Hank平衡盐溶液5.3mMCa2+PH7.4编解KCl(5.4mM)、NHCO3(4.2mM)、NACl(136.9mM)、D-Glucose单水合物(5.6mM)(全部从Kemig购买)、KH2PO4(0.4mm克米卡,萨格勒布,克罗地亚) Na2HPO4x2H2OFluka ChemieAG,Buchs,Service)和Cacl2x2H2O(5.3mmM)优化微镜/manitol粉混合显示有利的生物药理和喷雾性能Nasal沉积研究揭示策略的潜力 高效DSP交付所得结果为扩展研究为最终验证所需要适当的维夫模式提供了坚实基础。