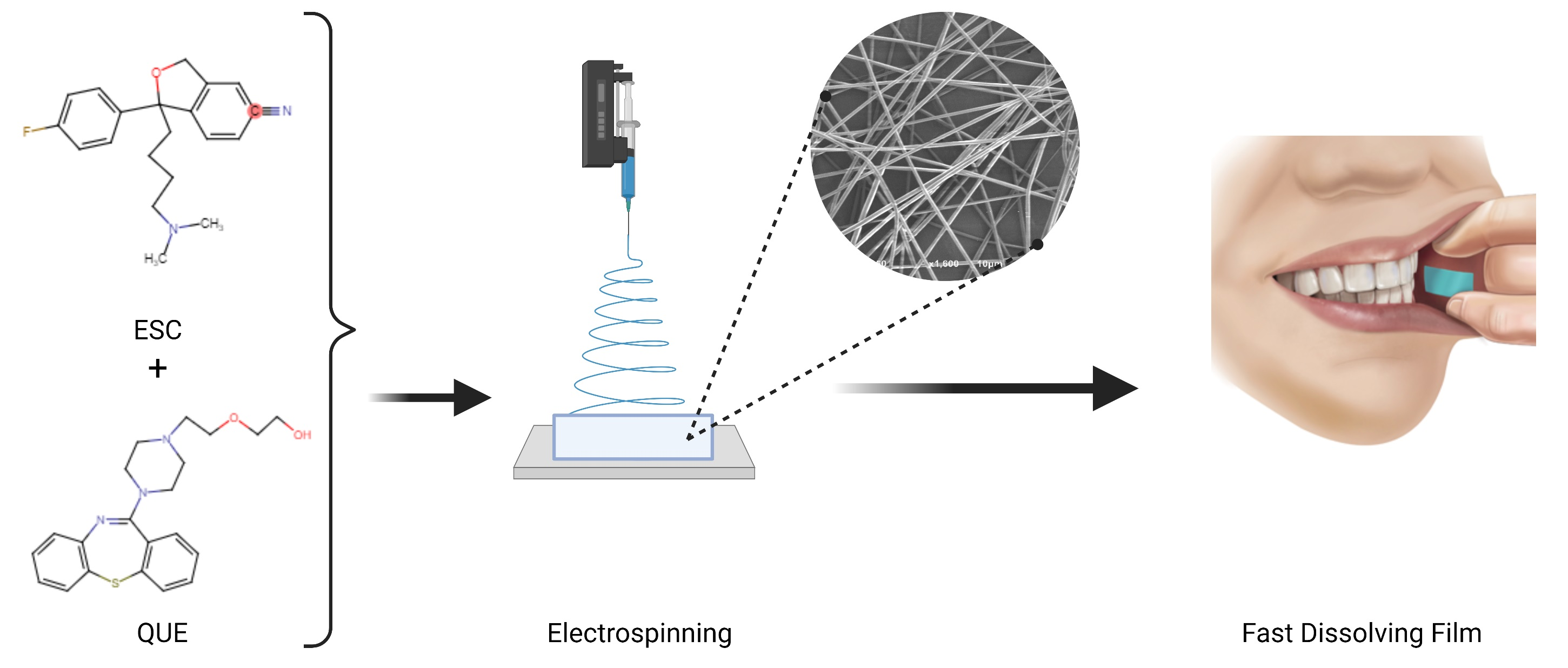
Major depressive disorder (MMD) is a leading cause of disability worldwide.大约三分之一的MDD病人未能实现响应或释放,导致抗药性抑郁症心理药理策略之一 克服tRD综合使用抗心理药剂加选Serbonin复用抑制器包括sitalopram/Quetiapine处理重大抑制症 erschenzuersta

Major depressive disorder (MMD) is a leading cause of disability worldwide.大约三分之一的MDD病人未能实现响应或释放,导致抗药性抑郁症心理药理策略之一 克服tRD综合使用抗心理药剂加选Serbonin复用抑制器其中包括非典型反心理学克特平纳和sSI-escitalopram编译成定片组合状并用同轴电脉冲快速分解膜后发纤维形态学研究.
sem图片显示,药载纤维平滑、非测值和非浮度直径为0.9+0.1m,TEM图像则展示核心和外壳特殊层,确认这些纤维成功编译差分扫描卡路里和X射线分解研究确认,这两种药均在装药纤维内不定分布药载纤维分解时间为2s,加速释放两种药(5分钟后50%),使之成为口服粘合剂的诱人配方ex vio渗透性研究表明QUE渗透布氏膜,但不是ESC可能受缓冲上和细胞间脂阻塞Overall, the developed coaxial fibers could be a potential buccal dosage form that could be attributed to higher acceptability and adherence among vulnerable patients, particularly mentally ill patients.Download the full article as pdf: Fabrication and Characterization of Fast-Dissolving Films Containing Escitalopram, Quetiapine for the Treatment of Major Depressive Disorder
or read it here
Materials:
Polyvinylpyrrolidone (PVP!Mw=1300kDa/99.5%/99.5%/99.5NACl/subjectic Nasu2 2/HPO 4 <{99.0%/100.5%/990-100.5%/Louis,MO,USA),HPLC级Acetorile从PanReac应用HemITW试剂中获取(西班牙巴塞洛纳),Esitalopramexate (ESC) (999.95%) 和quetiapinefumate (QUE) (999.13%)由Saudi药工医疗应用公司(SPIMACOADWAIH)(沙特阿拉伯里雅得)慷慨捐赠分解水由MilliQ生成(Billerica,MA,USA),并用于编译模拟口水pH6.8和磷缓冲盐线pH7.4所有有机溶剂都高性能液相色谱学级。
alkahtaniM.E.欧达AH阿布阿萨布OA巴斯特AW欧鲁M陶菲克E.A.快速解析胶片编译/Quetiapine处理重大抑制症。 药理 2021 , 13 ,891https://doi.org/10.3390/pharmatutics130691
/azi
Starting from the premise that a reduced number of active pharmaceutical ingredients (APIs) are used to treat hypotension, the aim of this study consisted of developing new formulations of caffeine-orodispersible tablets (CAF- ODTs).配方变量类型分解和集中CAF-ODTs直接压缩编译

Starting from the premise that a reduced number of active pharmaceutical ingredients (APIs) are used to treat hypotension, the aim of this study consisted of developing new formulations of caffeine-orodispersible tablets (CAF- ODTs).配方变量类型分解和集中CAF-ODT直接压缩编译(CAF1、CAF2和CAF3)从药法学角度分析拟议配方配方开发平板外观、抗压性、易碎性、分解行为和体外咖啡因释放白片抗压下降下序CAF1>CAF2>CAF3易碎测试显示所有配方都尊重欧洲药典欧尔10)值小于1%的要求三种配方分解时间小于180秒,CAF2最小时间注册,Sodium StarchGlycolate体外释放研究发现99.9%以上咖啡因从所有三种分析配方释放通过调查一分钟后释放的咖啡因量可见最大释放量记录在CAF2配方中,SSG被用作分解物与CAF2相比,CAF2释放量在前5分钟CAF1配方使用crosmellose基于所得结果我们可以得出结论,所有三种配方都尊重药法自律要求The presence of SSG in the CAF2 formulation led to obtaining tablets with a reduced disintegration time in comparison to the other two formulations proposed in this study.
Download the full article here: Developing and evaluation of orodispersible tablets containing caffeine
More on CMC and Croscarmellose Sodium
Article Information: Ro J Pharm Pract.2021;14(1) DOI: 10.37897/RJPhP.2021.1.5 by Assist.师傅Robert-Alexandru Vlad,Elena-BeatriceTrifan讲师Paula Antonoaea师傅Nicoleta Todoran教授Adriana Ciurb
Developing orodispersible tablets with caffeine – Materials and Methods
To obtain orodispersible tablets the following pharmaceutical ingredients were used: API – caffeine – (CAF – Rochem, United States of America – USA), lactose, Lactopress® (LCT – DFE Pharma, Germany), sodium croscarmellose, Vivasol®CCS – JRS Pharma, Austria), sodium starch glycolate, Primojel® (SSG – DFE Pharma, Germany), Aerosil® 200 (Degussa, Japan), Magnesium stearate (Alfa Aesar, USA), mannitol, (MNT – VWR Chemicals, USA) and banana flavour (BFL – JinTai, China).所拟配方使用10毫米拳获取偏心按压器(Korsch,德国)混合粉末使用VBlender值(YM-4-United Kingdom)逆序密度,最后添加粉末为润滑油Three formulations of caffeine orodispersible tablets (CAF-ODTs) were produced noted as CAF1, CAF2 and, CAF3, their composition is presented in Table 1.
Der Beitrag Developing and evaluation of orodispersible tablets containing caffeine erschien zuerst auf Pharma Excipients.

Introduction: Tablets are still the most widely used dosage form for oral application due to their simple administration and relatively low manufacturing costs.片片的主要缺陷之一是吞咽或咀嚼难这一点对特殊病人群体特别相关,例如儿科和老年病人[1]避免问题并保留 [.]
DerBeitrag

Introduction: Tablets are still the most widely used dosage form for oral application due to their simple administration and relatively low manufacturing costs.片片的主要缺陷之一是吞咽或咀嚼难这一点对特殊病人群体特别相关,例如儿科和老年病人[1]为了避免问题并仍然允许口服药理疗,可使用可分片法可分辨片片快速分解 后他们的管理口不嚼或进水在ODTs帮助下易用液体用量表可与固态药用表高物理和化学稳定性相容[2]调查重点是直接压缩协同处理前接受者,并用enalpril阳性、ibrofen和parecatamol作为API模型最理想的是,协同处理引出多功能前接收者,从而在足够强度和所需分解时间之间求得更好的平衡[3]这项研究的目的是比较两个新协同处理前接受者与商业上已有的接受者。For this purpose, the dependence of disintegration time on achieved tablet strength was analyzed for seven CPEs including each of the three APIs.
Materials and methods: The CPEs investigated in this study were Granfiller-D®211 (Daicel, Japan), Hisorad® (Daicel, Japan), SmartEx®QD-50 (Shin-Etsu, Japan), Ludiflash® (BASF, Germany), Prosolv® ODT (JRS Pharma, Germany), Pearlitol® Flash (Roquette, France) and Parteck® ODT (Merck, Germany).每项测试CPE单选3模型API15分钟第二混合步1%添加为润滑剂并混合3分钟分批装有parecatamol和ibrofen(均50%)的火药混合模型APIs直接压缩在压缩模拟器Styl'One演化上(Medel'Pharm,法国)。CEP对低剂量配方能影响内容一致性(未显示)的程度进行彻底评价时,含有enalpril阳性混合物(4%)直接压缩在旋转平板按Korsch XM12上(Korsch,德国)。应用六种不同的压缩压力(50-250兆帕)。使用直径9毫米的扁平直拳表格属性用SmartTestST50测量(瑞士Soxat)。抗拉强度根据Fell和牛顿计算Disintegration studies were performed in a disintegration test apparatus (Z32 Erweka GmbH, Germany) according to Ph.Eur.10.
 Download the full poster as a PDF here
Download the full poster as a PDF here
Article information: M.科科特Okabayashi A卢拉市维德伊市布赖特克鲁兹药理学和生物药理学学院Heinrich Heine大学40225 Desceldoration11823Konan直接归并Orally分解表宽度=1170自动机游戏clipboard-write!encrypted-media!陀螺仪图片中允许全屏++/iframe++/p

There is strong clinical need for pediatric formulations from healthcare professionals and caregivers since the number of available pediatric formulation is limited.当前口语管理是市场儿科配方的主要管理路线开发儿科口服管理时,应主要考虑“可接受性”和“处理性”。具体说来,ss//pharmaexccepties.com/news/application-mini-odts-pediacrics/

There is strong clinical need for pediatric formulations from healthcare professionals and caregivers since the number of available pediatric formulation is limited.当前口语管理是市场儿科配方的主要管理路线开发儿科口服管理时,应主要考虑“可接受性”和“处理性”。特别重要的是配方,因为它包括可达性(如品味、嗅觉和纹理)和吞咽性(如尺寸和形状)。这两项因素决定了向儿童服药的适切性(图二图二i/p>
液剂量表可以取自新生儿阶段,但有几个问题。与固态配方相比,它们的存储条件和储存寿命受限一瓶覆盖数日药量,护理员必须测量液度,难精确使用单剂量各种固态用量形式,如平板药、胶囊和粉末其中包括口服分解片片片片表,它是一种用量表,旨在提高传统片片片的吞化性,增加药的耐用性。
ODTs分解口服口服口服口服口服口服30秒内口服口服口服口服口服口服口服,口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服,口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服口服ODT的优缺点包括安全性(他们预防期望)、方便性(时间和地点)和可靠性(他们防止拒绝服药)。然而,似可认为常规ODT快速分解并不足以供儿科使用。文章讨论特殊用量表,以便从前台制造厂家的角度提高儿科配方的可接受性。granflier-dg-d215目标s.com/product/granfer-gnf-d215使用专有粒子法制作特征用Fig表示二叉两种产品都提供优势,如提高ODT性能、缩短开发时间和简化制造过程Using these co-processed excipients, we developed ODTs which shape are suitable for pediatric use.
Download the full article as a PDF here
Source: DAICEL
See also our video on Granfiller-D here:
HiSORAD is composed of three compendial grade excipients, D-Mannitol, Microcrystalline cellulose and Croscarmellose sodium.
Characteristics
- Excellent compactibility
- Well-balanced tablet property between OD time and hardness
- High API loading capacity
- Simple composition
More on DAICEL ODT excpient products here