
The current study deals with formulation and evaluation of nateglinide solid dispersion (SD) incorporate into tablet formulation for controlled release of the drug.方法:NateglinideSD使用crospovidone编译并评价药含量和药分解优化自定义配方用HPCCK100和Cederela口香糖嵌入平板理表板配方都
DerBeitrag

The current study deals with formulation and evaluation of nateglinide solid dispersion (SD) incorporate into tablet formulation for controlled release of the drug.
Methods: The nateglinide SD prepared using crospovidone and evaluated for drug content and drug dissolution.优化自定义配方用HPCCK100和Cederela口香糖嵌入平板所有片状配方评为压缩前、压缩后、药分解和前受体兼容性研究。
Conclusion: The combination of SD and application of hydrophilic and hydrophobic polymers in matrix formation facilitated superior dissolution and absorption profile with greater patient compliance.
Sunitha, et al.: Development, characterization of nateglinide SD, and incorporation into tablet formulation for controlled release of drug – Asian Journal of Pharmaceutics • Jan-Mar 2021 • 15 (1) | 122 – Download full article here: Design, Development, and Evaluation of Controlled Release Tablets of Nateglinide Solid Dispersions
MATERIALS AND METHODS
The drug nateglinide, HPMC K 4M, HPMC K 15M, HPMC K 100M, and crospovidone were gifted from Hetero drugs Ltd, Hyderabad.The excipients Soluplus, Kolliphor ELP, and Kolliphor RH40 were purchased from Gattefosse, Mumbai.Lactose,talcsylpriodone (/div>
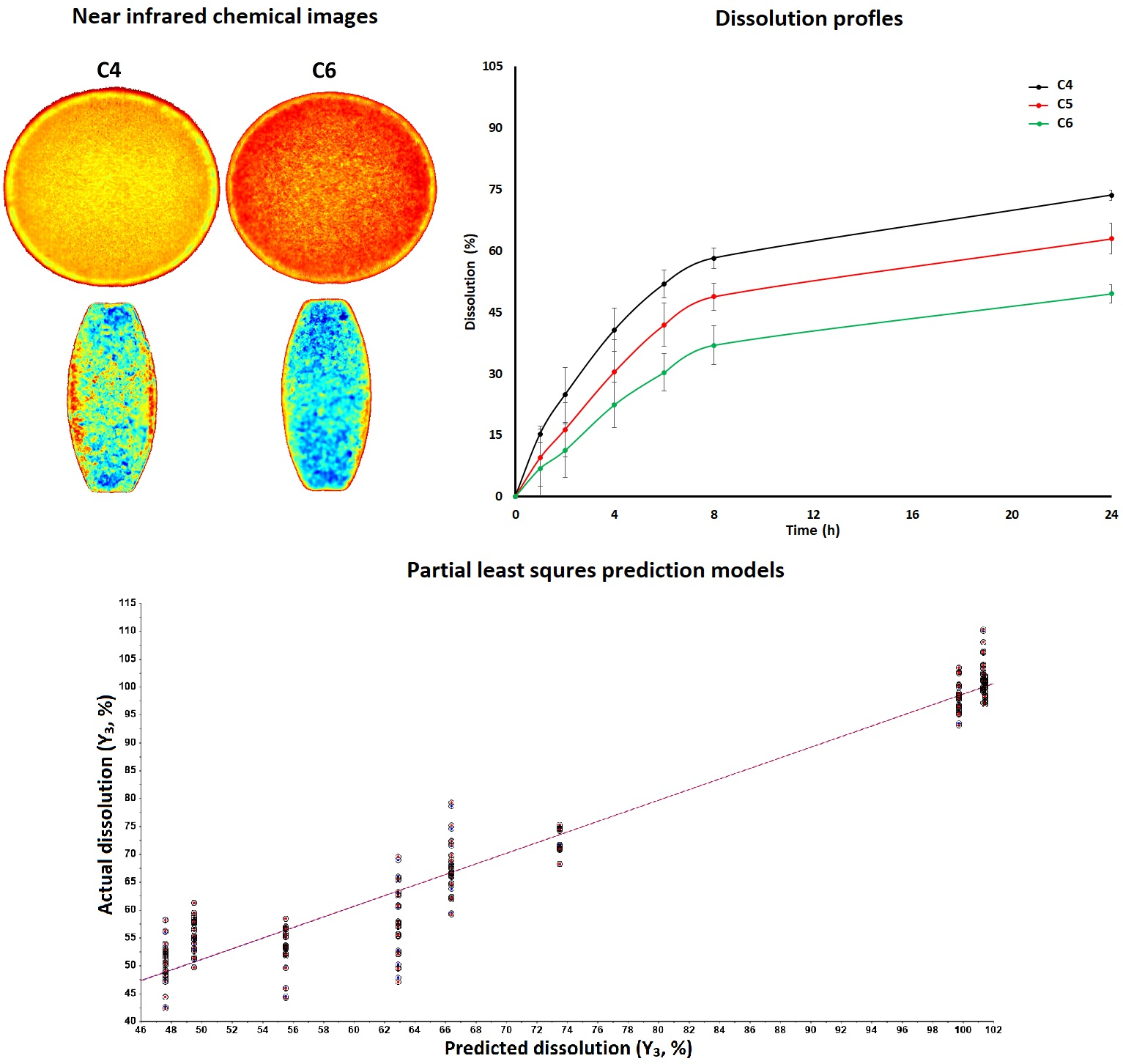
The focus of the present investigation was to develop a predictive dissolution model for tablets coated with blends of cellulose acetate butyrate (CAB) 171-15 and cellulose acetate phthalate (C-A-P) using the design of experiment and chemometric approaches.diclofenac钠被用作示范药叠加权重增量(X1、5、7.5和10%)和CAB [...]
Der Beltrag The focus of the present investigation was to develop a predictive dissolution model for tablets coated with blends of cellulose acetate butyrate (CAB) 171-15 and cellulose acetate phthalate (C-A-P) using the design of experiment and chemometric approaches.diclofenac钠被用作示范药叠加权重增量(Xsub>1 < 5,7.5和10%)和CAB171-15百分数(Xsub>2 3,50和66.7%)受监控响应分解时间为1(Y 1 )、8(Y 2 )和24(Y 3 p < 0.05]对Ysub>1 和Xsub>2 对Ysub>1 1 、Y 2、 和Ys3 1 、Y 2 和Ysub>sub>1 2、sub>3 预测独立样本值接近实际值最后,可以预测用两种方法混合细胞酯片片片的解析. 
Der Beitrag Development of a Multivariate Predictive Dissolution Model for Tablets Coated with Cellulose Ester Blends erschien zuerst auf Pharma Excipients.

The extensive use of oral dosage forms for the treatment of diseases may be linked to deficient pharmacokinetic properties.在有些情况下药难解而在别体中,配方快速传递肠道令生物实现治疗水平moreover, some drugs must act […]
Der Beitrag In-Depth Study into Polymeric Materials in Low-Density Gastroretentive Formulations erschien zuerst auf Pharma Excipients.

The extensive use of oral dosage forms for the treatment of diseases may be linked to deficient pharmacokinetic properties.在有些情况下药难解而在别体中,配方快速传递肠道令生物实现治疗水平某些药因胃病理必须在当地作用,使用能改善所有参数并增加宿主时间的配方已成为多项研究工作的目标,低密度系统最有希望并广博探索,然而,还有进一步改进这些系统的余地。低密度催化系统使用各种聚合材料并用多种方法提高药物生物可用性This works aims to expedite the development of breakthrough approaches by providing an in-depth understanding of the polymeric materials currently used, both natural and synthetic, their properties, advantages, and drawbacks.
Download the full publication here: In-Depth Study into Polymeric Materials in Low-Density Gastroretentive Formulations
Download the supplementary materials here: Supplementary Materials- In-Depth Study into Polymeric Materials in Low-Density Gastroretentive Formulations
or read it here: https://www.mdpi.com/1999-4923/12/7/636#cite
Iglesias, N.!加尔比斯ERomero-Azugle L.贝尼托ELucas RGarcía-MartínM.G.de-PazM-Vs/www.mdpi.com/1999-4923/7636#cite目标s药理2020,12,636.
Der Beitrag In-Depth Study into Polymeric Materials in Low-Density Gastroretentive Formulations erschien zuerst auf Pharma Excipients.
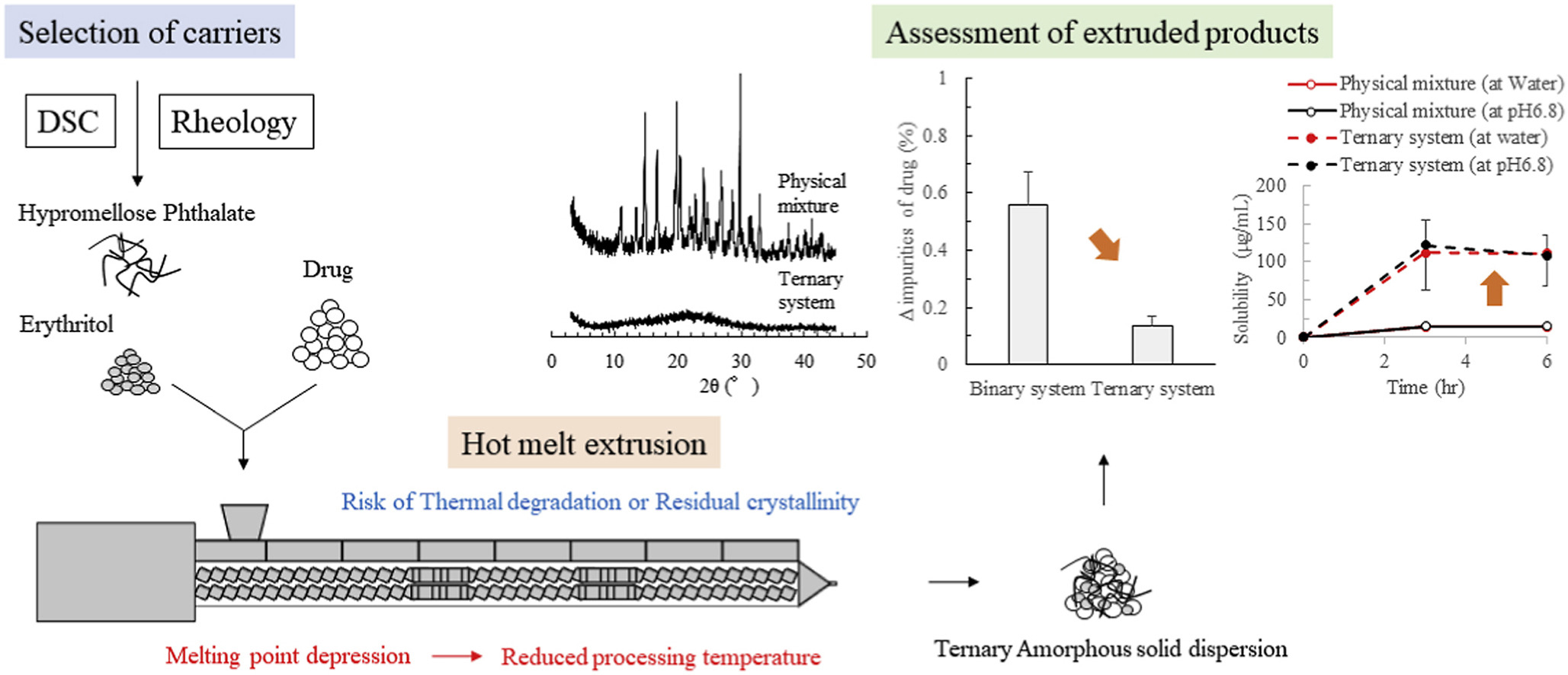
Hot melt extrusion is a promising technology for producing amorphous solid dispersions, however, there is a risk of thermal degradation or residual crystallinity formation.可优化配方和制造条件解决这一警告问题。研究的目的是探索优化配方方式,评价热动和热风学属性 [.]
Der Beitrag
Hot melt extrusion is a promising technology for producing amorphous solid dispersions, however, there is a risk of thermal degradation or residual crystallinity formation.可优化配方和制造条件解决这一警告问题。研究的目的是通过评价热动和热Rhelogy特性探索优化配方,与各种载体混合并发并并调查所得系统效果griseofulvin与14种聚合物、1dapride或4种糖醇结合使用差分扫描卡路里和Rhelogy分析此外,HP橡胶状态与Ery混合时保持广温范围极有希望候选配方使用双螺旋exruder制造装有HP和Ery的所有稀释产品显示热降解下降此外,产品显示提高溶性并保持超饱和富集度,与晶状磁性、物理混合或与HP二元配方生成的挤压产品相比Continue here
Keywords Twin screw extruder, Differential scanning calorimetry, Rheology, Thermal degradation, Residual crystallinity, Parteck, Parteck MXP, Kollidon VA64, Kollicoat IR, Pearitol 200SD, Xylisolb 700, Parteck SI, Carbopol 971PNF, Metolose SM-4
Der Beitrag Characterization of ternary amorphous solid dispersion containing hypromellose phthalate and erythritol prepared by hot melt extrusion using melting point depression erschien zuerst auf Pharma Excipients.
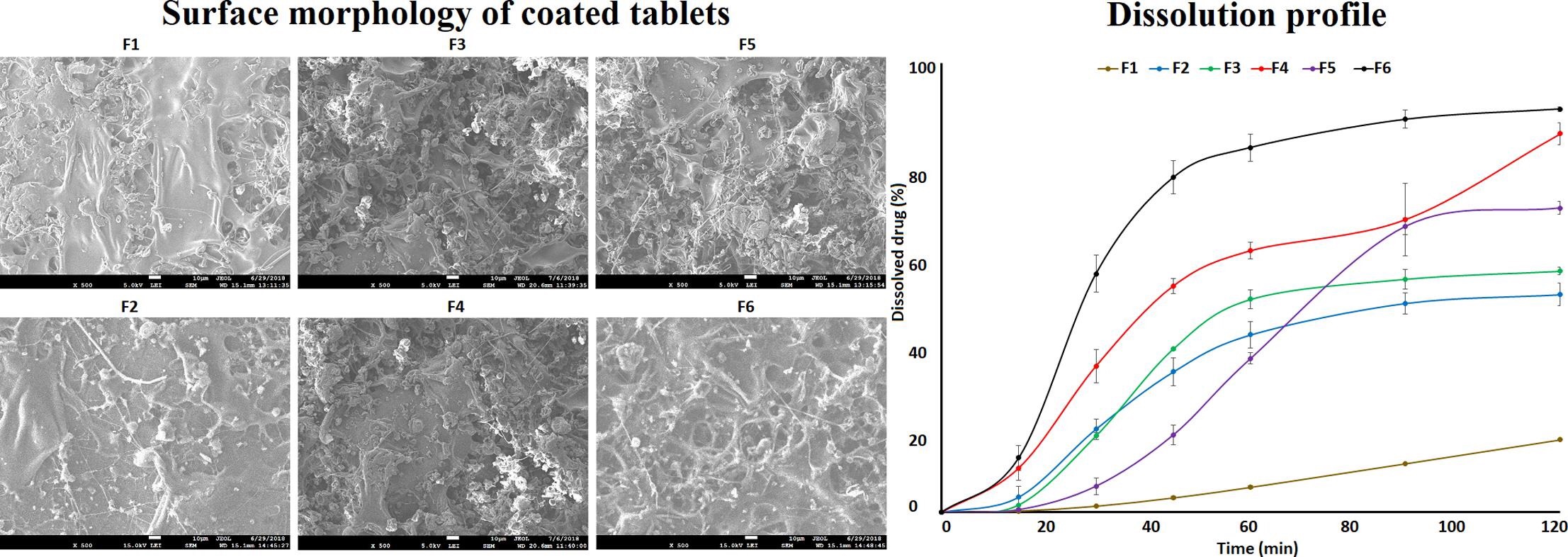
The focus of this work was to explore feasibility of using blends of cellulose esters (CA 320S, CA 3980-10 or CAB 171-15) and enteric polymers (C-A-P, Eudragit® L100 or HPMCP HP-55) for delayed and enteric coating of tablets containing either diclofenac sodium (DFS, high dose) or prednisone (PDS, low dose) drug.核心药片 < p>DerBeitrag < arel=s/www.pharmaex新手s.com/news/blend-of-cellulose-ester-entic-聚合物-for-de

The focus of this work was to explore feasibility of using blends of cellulose esters (CA 320S, CA 3980-10 or CAB 171-15) and enteric polymers (C-A-P, Eudragit® L100 or HPMCP HP-55) for delayed and enteric coating of tablets containing either diclofenac sodium (DFS, high dose) or prednisone (PDS, low dose) drug.
The core tablets of DFS or PDS were coated with polymer blends to achieve approximate weight gain of 5% and 10%.涂片特征解析(0.1NHCl和磷酸缓冲pH6.8)和表面形态CA398-10或CAB171-15聚合物混合体表面形态粗糙易碎不到0.5%的药分120分解酸相解测试中5% w/w封片片分解.
PH6.8介质解析从16.2+0.2-98++2.1%和30.1+++0.5%-101.7+3.4%不等,分别来自DFS和PDS涂片分解CA320S混合比CA398-10或CAB171-15混合磷酸此外,在DFS或PDS封装片片片解析方面没有观察到重大差异。药用量可以解释Additionally, dissolution was higher in tablets coated with enteric polymer alone compared with the blends.In conclusion, core tablets can be coated with cellulose ester and enteric polymers blend to impart both delayed and enteric release feature to the tablets containing hydrophilic or hydrophobic drug. Continue reading
Der Beitrag Blend of cellulose ester and enteric polymers for delayed and enteric coating of core tablets of hydrophilic and hydrophobic drugs erschien zuerst auf Pharma Excipients.
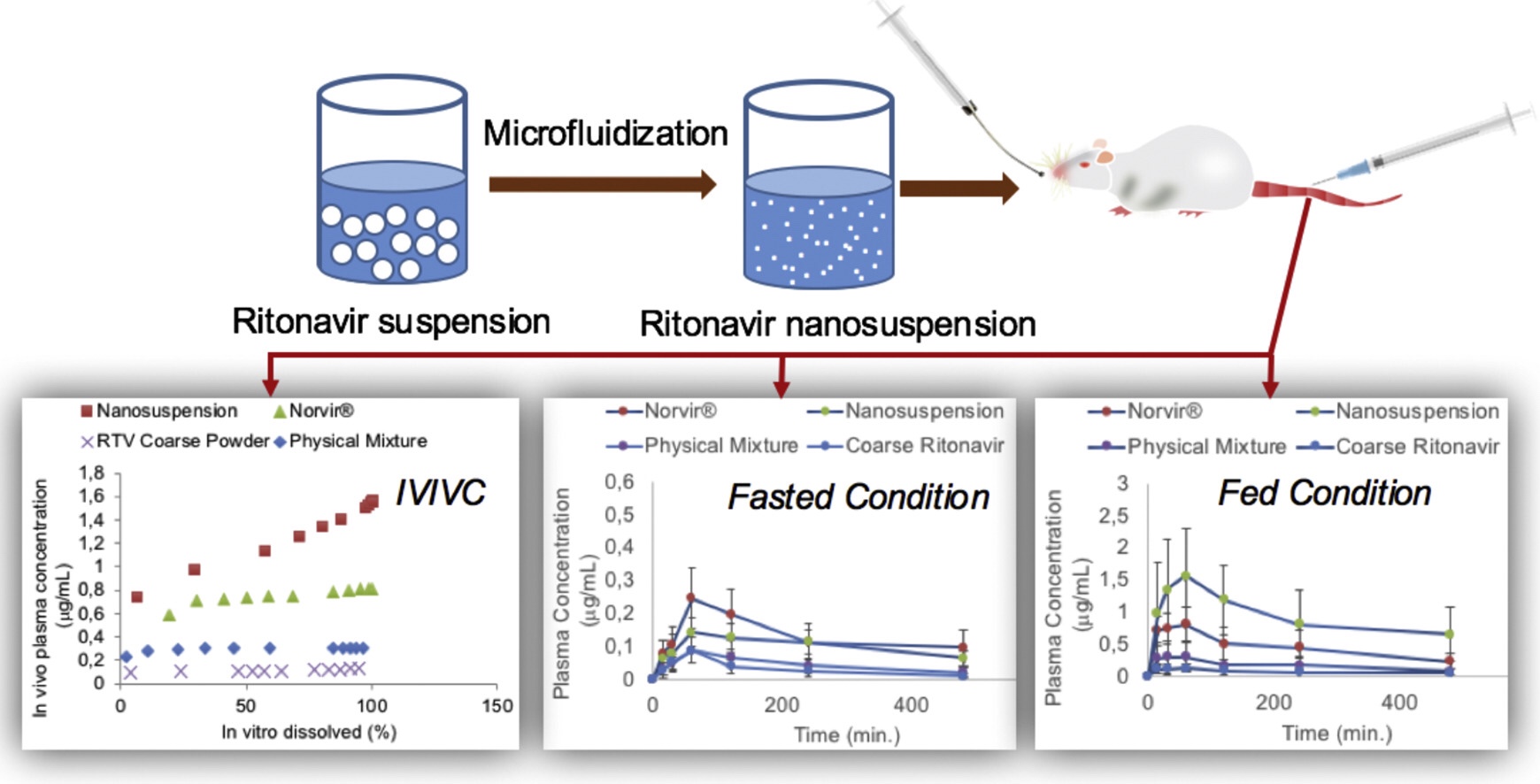
The main objective of this study was to evaluate the pharmacokinetics of ritonavir (RTV) nanosuspension in rats in both fed and fasted state in comparison with coarse powder, physical mixture and commercial product (Norvir®).点对点关系模型生成于体外分解结果和Vivo药效学研究结果The […]
Der Beitrag Evaluation of improved oral bioavailability of ritonavir nanosuspension erschien zuerst auf Pharma Excipients.

The main objective of this study was to evaluate the pharmacokinetics of ritonavir (RTV) nanosuspension in rats in both fed and fasted state in comparison with coarse powder, physical mixture and commercial product (Norvir®).点对点关系模型生成于体外分解结果和活体药效研究结果之间。
纳米粒度540-550纳米、粒度分布0.1-0.4和兆兆维根据大鼠药理学研究显示,纳米悬浮Cmax值和AUC0-t值显示比粗粉增加8.9倍和12.5倍,比填充组比商业产品分别增加1.9倍和2.1倍点对点关系模型显示相关模型意义重大得出纳米悬浮是一种大有前途的药送系统,可提高Ritonavir口服生物可用性Continue here on oral bioavailability
Der Beitrag Evaluation of improved oral bioavailability of ritonavir nanosuspension erschien zuerst auf Pharma Excipients.
Implication of Quantitative Selection of Each Excipient in Product Development Excipients' role in designing different dosage forms does not require any introduction.以上这些添加物与药理活性物质并发增加这些特性的主要目的是增加大数配方并传递所期望的[.]
Beitrag Excipients' role in designing different dosage forms does not require any introduction.以上这些添加物与药理活性物质并发增加这些特性的主要目的是增加大片配方并传递期望属性始发者像药物一样,需要验证和标准化下一章简单介绍不同用量表使用前接收者,包括固态用量表、液态用量表和半固态用量表计药先进配方领域的持续开发,我们还覆盖纳米化前接收者。 
Implication of Quantitative Selection of Each Excipient in Product Development
Der Beitrag Current Developments in Excipient Science – Implication of Quantitative Selection of Each Excipient in Product Development erschien zuerst auf Pharma Excipients.

The use of natural polymer as excipients in pharmaceutical sector is expanding day by day.低成本安全分解是导致它们不同于其他来源的主要原因自然源有多种种类和特征药产品中可以多用作为前接收者为理想目的服务[.]
Beitrag

The use of natural polymer as excipients in pharmaceutical sector is expanding day by day.低成本安全分解是导致它们不同于其他来源的主要原因自然源有多种种类和特征药产品中可以多用作为前接收者为理想目的服务文章的目的是透视自然聚合物应用问题。
Beitrag < a rel='nof'href='https//www.pharmaextips.com/organic-stemps/aplial-sugars/
This work aimed to map and discuss tablet-coating choice, quality deviations of this process, and corrective actions taken by Research and Development (R&D) teams from pharmaceutical industries.在巴西对聚合胶片涂层进行了跨段研究:基于文献资料向数家公司研发分解寄送问卷 [.]
Beitrag
This work aimed to map and discuss tablet-coating choice, quality deviations of this process, and corrective actions taken by Research and Development (R&D) teams from pharmaceutical industries.
Methods
A cross-sectional study of polymeric film coating was conducted in Brazil: a questionnaire based on literature information was sent to R&D divisions from several different companies that produce coated tablets in the country, which focused on the main type of coating systems used, most common quality issues, corrective action to each nonconformity, and the main influences in the choice of the coating system.
Results
The most used film coating systems are hydroxypropylmethylcellulose + Polyethylene glycol (HPMC/PEG) and polyvinyl alcohol + PEG (PVA/PEG), and the most common non-conformities are orange peel, picking sticking, chipping, and peeling.主要的教程校正动作包括温度控制、泛速、喷雾和压力率所有结果都由我们根据文献和作者的专业知识分析稳定性是影响涂层系统选择的主要因素 。
+get>Clubation href=s/www.pharmaexemists/cellulose-sters/qual-deviation-

The pressure to innovate faster than their competitors has drug formulators exploring new ways to optimise manufacturing processes Article in manufacturingchemist.com Continuous manufacturing, for example, has evolved at a relatively fast pace, leaving drug manufacturers feeling equally challenged and encouraged to evaluate their current production methods and adopt new technologies.端端结果为 ss/www.pharmaexccepties.com/organic-stemists/cellulose-esters/the-rise-of-exccepties/

The pressure to innovate faster than their competitors has drug formulators exploring new ways to optimise manufacturing processes
Article in manufacturingchemist.com
Continue reading on independent excipients in manufacturingchemist.com
Der Beitrag The rise of independent excipients erschien zuerst auf Pharma Excipients.

Oral modied-release multiparticulate dosage forms, which are also referred to as oral multiple-unit particulate systems, are becoming increasingly popular for oral drug delivery applications.聚合叠加多片生成持续释放剂量表比硬胶囊优先多片位解决慢性病和病人坚持问题大有希望 [.]
DerBeitrag Oral modied-release multiparticulate dosage forms, which are also referred to as oral multiple-unit particulate systems, are becoming increasingly popular for oral drug delivery applications.聚合叠加多片生成持续释放剂量表比硬胶囊优先并分片片片子大有希望解决慢性病、病人坚持症和吞入二分法问题,如果嵌入可分解矩阵中的话。多段压缩往往损害功能聚合物大衣,导致快速释放药物并继而丧失持续释放性能本审查向前端关键配方变量带入,这些变量有可能使包装多片压缩成持续释放片片重点是涂药粒子、微粒子和纳米粒子平板化,每个粒子都有一个专用段Furthermore, it explores the various approaches that are used to evaluate the compaction behaviour of particulate systems. Download full mdpi.com article: pharmaceutics-10-00176.pdf Der Beitrag Review Oral Modied Release Multiple-Unit Particulate Systems: Compressed Pellets, Microparticles and Nanoparticles erschien zuerst auf Pharma Excipients.

Physical and rheological properties of agglomerated xanthan gum (XG), commonly used as a food thickener for the management of the patients with dysphagia (swallowing difficulty), were investigated at different concentrations (0, 2, 4, and 6% w/w) of hydroxypropyl methylcellulose (HPMC) as a binder in the fluidized bed agglomeration process.s/www.pharmaex新手/cellulose/phal-and-rhelogic-properies-of-xanthan-gum-grumerate-bed-bed-eff-hpmc-a-binder/

Physical and rheological properties of agglomerated xanthan gum (XG), commonly used as a food thickener for the management of the patients with dysphagia (swallowing difficulty), were investigated at different concentrations (0, 2, 4, and 6% w/w) of hydroxypropyl methylcellulose (HPMC) as a binder in the fluidized bed agglomeration process.聚积XG粉流特征使用Carr索引和Hausner比评估HPCC绑定器获取的聚积XG粉比聚积无绑定物粉的易流性强多孔性强聚积XG粉末动态模Li聚积化粉末与HPCC绑定值远比没有HPCC聚积化粉末值低,表明它们的弹性性能因HPCC绑定器增加而增强hpc/content/uploads+8/10/1-s2.0-s0141114-ma
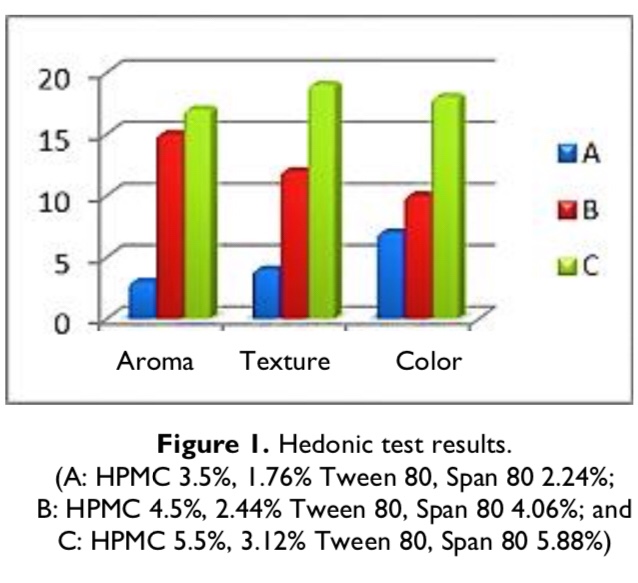
ABSTRACT Acne is the condition of abnormal skin which is indicated by inflammation cause by the bacterial infection of Propionibacterium acnes.自然线用于acne医疗achasin蛋白有抗菌活动slipse-sule-sliftina-a

ABSTRACT
Acne is the condition of abnormal skin which is indicated by inflammation cause by the bacterial infection of Propionibacterium acnes.自然线用于acne医疗achasin蛋白有抗菌活动螺旋粘膜制作成emulgel形式这项研究使用实验法,emulgel配方使用各种仿真剂和凝胶代理Tween801.76%、2.44%、3.12%、span802.24%、4.06%、5.88%和HPCC3.5%、4.5%、5.5%其它添加物有prileglycol、Metaben、bjylparben、parffin液化物、menthol和aquadest4周内物理评价测试这些配方,存储室温度、刺激测试、重音测试和循环测试染色体测试显示乳胶白色与奇味emulgel均同质非刺激性,水中含浸泡式油(o/w)。emulgel还满足pH值正常皮肤并传播能力范围emulgel粘度移位 < 10%,粘度值逆比传播能力公式C为3.12%Tween80,5.88%Span80和5.5%HPCC称为室温和循环测试后最稳定的公式It was also the most preferred by panelist.
Download Article here: 369-article-text-1402-1-10-20181010.pdf
Der Beitrag FORMULATION OF SNAIL SLIME (Achatina fulica) ANTI-ACNE EMULGEL USING TWEEN 80-SPAN 80 AS EMULSIFYING AND HPMC AS GELLING AGENT erschien zuerst auf Pharma Excipients.
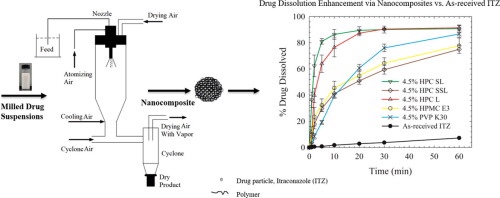
We explore the impact of various polymers and their molecular weight on the stabilization of wet-milled suspensions of itraconazole (ITZ), a poorly soluble drug, and its dissolution from spray-dried suspensions.ITZ悬浮SLSL、SLL和L级液化素(HPC)均分子权数为40、100和140k/mol

We explore the impact of various polymers and their molecular weight on the stabilization of wet-milled suspensions of itraconazole (ITZ), a poorly soluble drug, and its dissolution from spray-dried suspensions.ISL、SLL和L级液化素ITZ悬浮量分别为40、100和140k/mol,HPSS-SDS激光偏差结果显示2.5%HPCSL-0.2%SDS生成最佳ITZ纳米悬浮,而没有SDS,只有4.5%HPCSL/L级保证最小聚合神经特征显示汇总悬浮显示显性伪复性,而稳态悬浮显示牛顿行为喷雾干燥生成纳米复合物60-78%表示ITZ加载和可接受内容一致性Severe aggregation occurred during milling/drying when 4.5% polymers with MW ≤ 50 kg/mol were used!纳米复合体显示不完全再分解,原因是矩阵缩放缓慢,并在分解测试中缓慢释放ITZ高药载量免冲ITZ纳米编译器显示立即释放(>80分解20分数)时,通过喷雾驱动ITZ4.5%HPCSL/L.
/ahref='https://www.scient.com/science/article/pii/S092188182689>粒子增密度取决于各种配方和过程变量,包括关键配方前接收器的物质属性,如绑定器已知绑定物属性对微粒生长速度和广度的影响 [.]
derBeitrag 粒子增密度取决于各种配方和过程变量,包括关键配方前接收器的物质属性,如绑定器已知绑定物属性对微粒生长速率和广度的影响和湿粒化过程的密度可以是特定或不可知性对特定活性药素成份或药产品配制绑定属性和流程变量(如加法模式)对HSWG进程和产品性能的影响可以实验式探索或通过硅建模和模拟探索介绍实例和案例研究使用实验法和建模法对过程性能的影响This chapter demonstrates the essential role of combining multiscale models such as the mechanistic, first principles-based particle-level models, and process models with experimental observations to fully understand and characterize the role of binder attributes on the process outcomes. Read more on wet Granulation in the Handbook of Pharmaceutical Wet Granulation Der Beitrag Effect of Binder Attributes on Granule Growth and Densification erschien zuerst auf Pharma Excipients.
