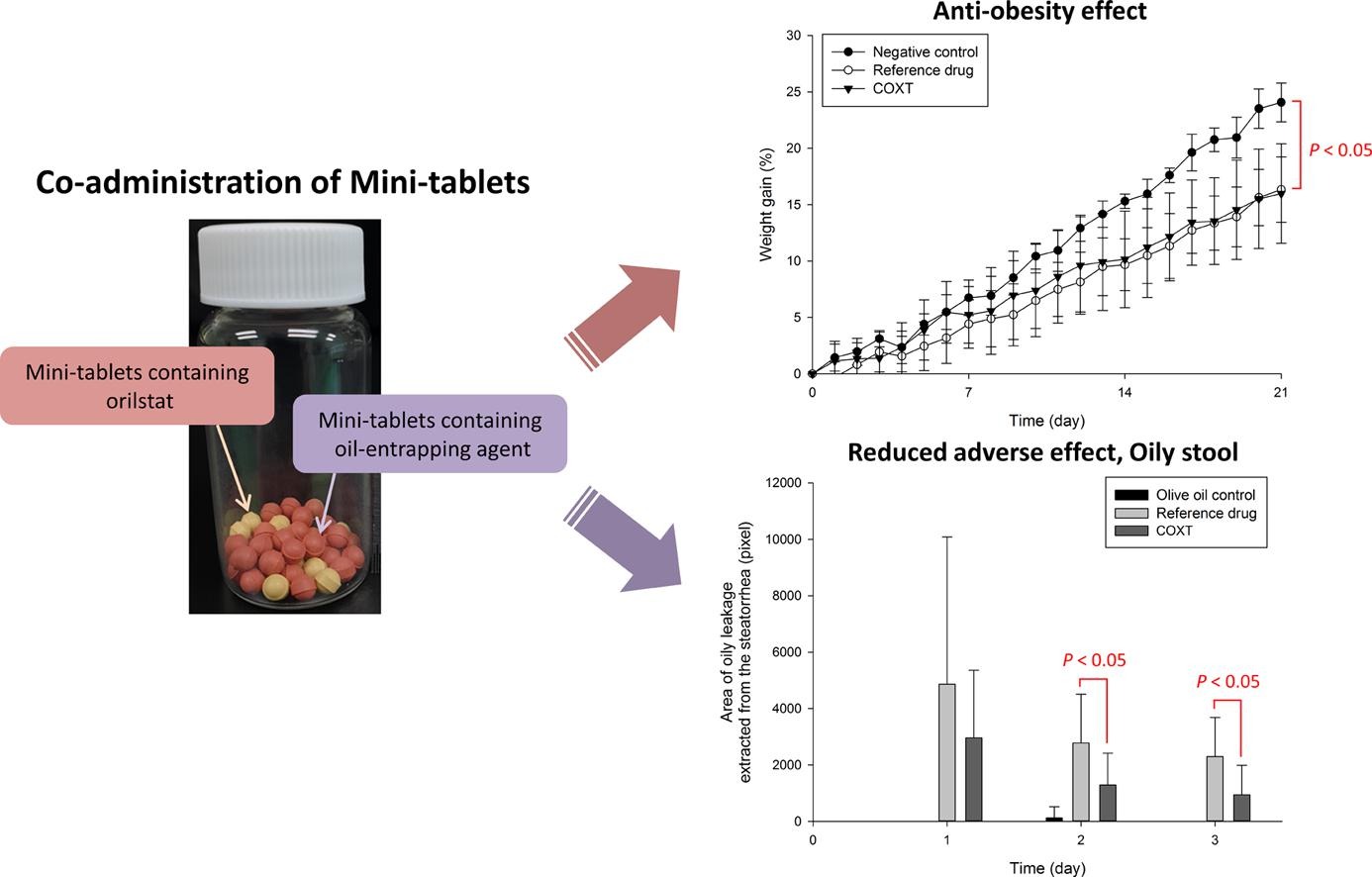
The purpose of this study was to develop an oral dosage form of orlistat for the treatment of obesity with reduced adverse effects, for example, fatty and oily stool that have been reported to be associated with the mechanism of action of orlistat.基于本研究所得体外结果,Xanthan口香糖被选作加油代理因此,建议对含有单片和含有单片口香糖的小片进行联合管理,以优化分片表或单片表准备微型目录显示等效药物释放剖面值, f 2 ,50多与商业营销或立即释放胶囊Xenical此外,优化配方还显示活性反渗透效果类似于Xenical+Q具体地说,Sprague-Dawley大鼠排出粪便分析显示,优化配方比XenicalCaps(P WSR coagulant, sodium carboxylmethyl cellulose, microcrystalline cellulose (MCC-101, Ceolus 250HHX pharm), polyacrylic acid (PA, Carbopol® 974P), chitosan, colloidal silicon dioxide (SO, Aerosil® 200), magnesium aluminium silicate (MAS, Neusilin® US2), glyceryl behenate (GB, Compritol® 888 ATO), glycerol monooleate (GMO, Peceol
250HHX pharm), polyacrylic acid (PA, Carbopol® 974P), chitosan, colloidal silicon dioxide (SO, Aerosil® 200), magnesium aluminium silicate (MAS, Neusilin® US2), glyceryl behenate (GB, Compritol® 888 ATO), glycerol monooleate (GMO, Peceol ), polyoxyl 40 hydrogenated castor oil (CO, Kolliphor® RH 40),Kolliphor® RH 40macrogolglyceride (CMG, Labrasol®), sodium starch glycolate (SSG, Primojel®), crospovidone (CP, Kollidon® CL), sodium lauryl sulfate, polyethylene glycol 400 ( d-α-tocopherol acetate (Vitamin E acetate,, polyvinylpyrrolidone (PVPK30, Kollidon® 30), magnesium stearate, potato starch
), polyoxyl 40 hydrogenated castor oil (CO, Kolliphor® RH 40),Kolliphor® RH 40macrogolglyceride (CMG, Labrasol®), sodium starch glycolate (SSG, Primojel®), crospovidone (CP, Kollidon® CL), sodium lauryl sulfate, polyethylene glycol 400 ( d-α-tocopherol acetate (Vitamin E acetate,, polyvinylpyrrolidone (PVPK30, Kollidon® 30), magnesium stearate, potato starch
Der Beitrag Anti-obesity effect with reduced adverse effect of the co-administration of mini-tablets containing orlistat and mini-tablets containing xanthan gum: In vitro and in vivo evaluation erschien zuerst auf Pharma Excipients.

 250HHX pharm), polyacrylic acid (PA,
250HHX pharm), polyacrylic acid (PA, 



 How to formulate a simple emulgel with a high level of Transcutol® P
How to formulate a simple emulgel with a high level of Transcutol® P