
Hot-melt extrusion has found extensive application as a feasible pharmaceutical technological option over recent years.HME应用包括溶性增强、口味掩码和持续释放毒品生物可用性增强是当今科学热题,HME的主要应用之一是非静态固态分布t-mel-Expression/

Hot-melt extrusion has found extensive application as a feasible pharmaceutical technological option over recent years.HME应用包括溶性增强、口味掩码和持续释放毒品生物可用性增强是当今科学热题,HME主要应用之一是非态固态散射量。
本审查描述HME技术最重要的方面及其用于准备固态散射量,作为提高易溶性药物溶性的一种药方配制策略springer.com/article/10108/s12249-021-7#citeas目标='blank'rel=nopener'Simosss热-MeltExpressive:产品开发路径图aspsPharmScitech 22, 184(2021年)。https://doi.org/10.1208/s12249-021-02017-7
See the new webinar about HME and ASD here:
Der Beitrag Hot-Melt Extrusion: a Roadmap for Product Development erschien zuerst auf Pharma Excipients.

The current study evaluated the effect of location and amount of various superdisintegrants on the properties of tablets made by twin-screw melt granulation (TSMG).单片胶片内分层使用可容性、紧凑性、压缩性以及易碎性、分解性能和分解性能得到评估scre-mel-granation/

The current study evaluated the effect of location and amount of various superdisintegrants on the properties of tablets made by twin-screw melt granulation (TSMG).单片胶片内分层使用可容性、紧凑性、压缩性以及易碎性、分解性能和分解性能得到评估异常加法导致快速分解和分解CPV性能优于CCS和SG微粒固片对CPV较低,但由于熔粒高塑变形,仅观察到可塑性微小下降语义内添加CPV导致延长分解剖面谱,这可能与分片期间孔隙损失相关百分数内添加CPV结果显著下降分解效率,而SSG性能不受粒子过程影响CCS不适合用于生产即时释放配方,当它以总比例添加到粒子相时,但其效率比CPV低减值最短分解(78s)和分解(sub>80 4.2分钟)用CPV外形实现CPV和CCS内部生成增分时间高层次应用500sap15分钟内,只有 sSG显示进程和位置独立分解性能 。
s/sqstrection-droductioni>APSPharmSciTech 22, 183(2021年)。https://doi.org/1208/s12249-021-02056-0Der Beitrag
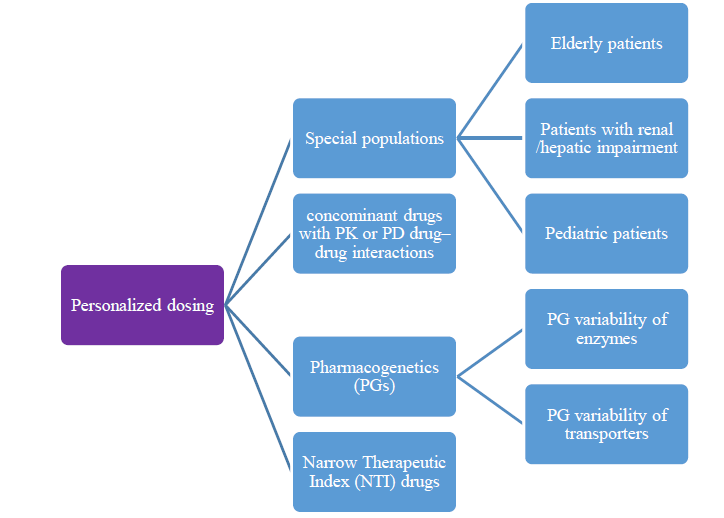
The oral route is the most common form of drug administration, being a safe and convenient, particularly for solid oral dosage forms.当前大规模制造技术大都产生固定剂量单元并允许有限选择用量强在某些情况下,需要用药强度个性化化,包括髓治法理学

The oral route is the most common form of drug administration, being a safe and convenient, particularly for solid oral dosage forms.当前大规模制造技术大都产生固定剂量单元并允许有限选择用量强在某些情况下,需要用药强度个性化化,包括髓治疗指数药或伴生药药药-药性/药效药-药性交互作用、酶或运输器药性变异性,以及特殊人群,包括老年人、儿童和肾功能缺陷/肝功能缺陷患者a类='btnbn-defaultbn-md'hrefs.com/wp-content/uploads/2021/06/this-print-a-aproach-to-Dose个性化可以通过复合或使用其他管理路径实现(例如侧接式),但这些路径缺少与标准制造相同的质量控制水平,不便/可接受性差不等。自动制造个性化固口化强力可解决这一需求开发三维打印自动逐层生成自定义平板三维打印片片子除单点使用外,还有可能用于早期临床试验期间的剂量升级、制片点药短期和制作复杂多药片片件并配有个人化配方图解特别是阻塞沉积建模(FDM),它涉及通过窄热喷嘴并随后沉积准备固态物体而分解热塑丝,似乎对临床现场生产有帮助,因为制造过程没有松散粉末,后处理可以避免。FDM应用需要FDM处理线程由热熔扩展制造。
IR是口服量表最常用发布剖面图,并同时产生来自通过热熔流程制造三维打印片片的固有特性的重大挑战 工作旨在为从FDM-和直接粉打印中产生的独特值建议创建选项,这些方面与标准制造过程基本不同(即分散化制作和定制平板结构)。临界质量属性(CQAs)集成为3D打印片片片的成功标准,包括内容一致性提供剂量精度(相对标准一致性数据偏差)、权重一致性(以确保内容一致性)、分解剖面貌(目标快速释放)和物理化学稳定性此外,还考虑了开发三维打印剂量表的可接受外观和大小。
关键目标之一是开发一个工业化配方工具箱,适合不同的API开发可缩热熔挤出配方并装有多种药量首项研究探索选定前接收者组合实现快速或极快从FDM打印片状带水益模型复合所选水溶聚合物和水分分解/处理辅助工具对FDM兼容性的影响,并结合填充密度对配方质量属性的影响(即重量和内容一致性、分解率)进行了研究。Hypray-Cellose SSL选择为水益聚合物和咖啡因,药载量5-20%热稳定模型药Poly-Serveridone-vilenegletate聚合物(在第三次研究中开发出易溶性模型复合立即释放FDM表,并使用非损性精确形态分析检测出分解关键结构参数基本methacrylate共聚物( 虽然FDM打印可能有利于临床现场制造,因为打印过程没有粉末或溶剂参与,但一大挑战是如何实现丝状所需机械性能允许FDM处理第四次研究探索直接粉色3D打印平板片,省略了简化引信沉积建模的丝片步骤直接三维打印时,粉状混合装入像盒头并成功打印出蜂窝设计跨步DPP内装孔度高表面积证明概念制作快速释放用量表水溶性HPCSSL选择矩阵前和咖啡因模拟药PEG4000增塑原型和KollidonQVA64快速分解聚合物对DPP可处理性和解析率的影响得到了调查3D打印低密度(30%)显示快速分解与配方无关,而高填密度(80%)则需要PEG4000和KollidonVA64组合实现快速释放所得平板显示药含量百分比完全一致,但有可变权重咖啡因以晶状状态和稳定多态形式出现直接粉末打印可行性即时释放用量表制作这种方法可创造避免热熔溢出的机会,允许三维打印独立于线性机械特性并有可能通过减热延长产品架存存存存期。
开发表板时对成人和儿童的尺寸都满足一致性内容需求,以提供精度剂量(相对标准一致性数据偏差)、权重一致性、分解目标(快速释放)和物理化学稳定性运算学研究表明,未来开发IR3D打印剂量表,除适当聚合矩阵选择外,还可以减少闭口孔量,增加开孔量,优化形状/大小和分解之间的平衡水益聚合矩阵与CAD设计相结合,通过FDM或DPP制造,对未来3D打印药产品开发大有希望然而,仍应克服的挑战包括直径整齐化丝状物、精度和可复制沉积质量、设计密度和实际密度差异、可预测最终形态学和丝状机械性能In addition, there might be a need in a real-time monitoring of flow and viscosity of the molten mass during the 3D-printing process.
Download the full dissertation as a PDF here
Author: Marina Fanous, Basel University.
Der Beitrag Three-dimensional printing of tablets – a potential approach to on-clinical-site extemporaneous formulation and personalized medicine erschien zuerst auf Pharma Excipients.

Following our study on the impact of hot melt extrusion (HME) process conditions on the product quality, we expanded our investigation to assessing the effect of scale-up on the product quality.arefs/www.pharmaexsubjects.com/priment-products-qual-hme/

Following our study on the impact of hot melt extrusion (HME) process conditions on the product quality, we expanded our investigation to assessing the effect of scale-up on the product quality.为此,我们研究流程设置和不同提升变异对实验植物级exruder有效药素退化的影响。
six扩展变异通过分析几个依赖过程变量并交叉参照实验室尺度外壳实验,我们辨识出某些模式The results of the reduced order mechanistic 1D HME simulation of various process states made it possible to establish a correlation between the achieved API degradation and the local melt temperature and the exposure time in specific zones along the screw configuration.
Since the same melt temperature and exposure time correlations were also valid for the lab scale-extruder, such an approach could be used in the future to predict the product quality as a function of processing conditions fully in silico prior to the first extrusion trials.
Download the full article as a PDF here or read it here
Article information: Josip Matić, Carolina Alva, Simone Eder, Kathrin Reusch, Amrit Paudel, Johannes Khinast.面向预测热熔溢出产品质量:实验工厂规模扩展,国际药理学杂志:X卷32021https://doi.org/10.1016/j.ijpx.2021.100084.
See a webinar about HME and ASD from one of the authors of this publication, Kathrin Reusch, here:
Der Beitrag Towards predicting the product quality in hot-melt extrusion: Pilot plant scale extrusion erschien zuerst auf Pharma Excipients.

In this series of webinars we will discuss the latest developments in the fields of amorphous solid dispersion (ASD) focusing on Excipient Key Attributes linked to the most popular technologies: Hot Melt Extrusion (HME), Spray Drying and their Downstream Processing.Shin-Etsu、Leistritz、ProCepT、Alexanderwerk和受邀讲演者会向您提供 [.]
Der Betrag In this series of webinars we will discuss the latest developments in the fields of amorphous solid dispersion (ASD) focusing on Excipient Key Attributes linked to the most popular technologies: Hot Melt Extrusion (HME), Spray Drying and their Downstream Processing. During these presentations Shin-Etsu, Leistritz, ProCepT, Alexanderwerk as well as invited speakers will give you more insights into ASD. Hot Melt Extrusion is a well established technology used in ASDs.除非溶解法的好处外,API和聚合物的某些属性必须像玻璃切换温度和分解温度一样加以考虑。 
FIRST WEBINAR – Tuesday 29 June, 3pm CEST, 9am EST
SECOND WEBINAR – Wednesday 22 September, 3pm CEST, 9am EST
THIRD WEBINAR – Thursday 28 October, 3pm CEST, 9am EST
First Webinar: Hot Melt Extrusion (HME)
The objective of this webinar is to address:
- How the extruder configuration can impact the ASD
- The advantages of HPMCAS in ASD
- How to optimize the extrusion process of HPMCAS
First presentation: Hot melt extrusion for amorphous solid dispersion – Process Design and Applications
Speaker:
Kathrin Reusch, Leistritz GmbH

Presentation abstract:
The presentation will give a short background on solid dispersion by Hot Melt Extrusion showing specific process layouts of marketed extruded products.
Based on the case study: "Hot Melt Extrusion of Ritonavir/Lopinavir" process relevant parameters and their impact on the final product quality including scalability will be discussed.
Speaker biography:
Kathrin obtained her degree as Master of Science at the University of Applied Science Fulda/Germany in 2014
The same year she joined Leistritz as a process engineer (Pharma) responsible for:
- Preparation, conduction and follow-up of extrusion trials – Definition of process layouts
- Process optimization, customer support & training
- Scale-Up experience
- Installation support (FAT, SAT)
- Organization of extrusion events (Pharma extrusion academy) and marketing tasks (website, publication, seminars)
In 2019 she took over the responsibility of product manager.
Second Presentation: Development of HPMCAS-based amorphous solid dispersions by melt extrusion
Speaker:
Abu Serajuddin, PhD, Professor of Industrial Pharmacy, College of Pharmacy and Health Sciences, St.John's University, Queens, New York, USA

近些年营销的7个含HPMCAS单片中,只有1个由HME制造HME不常用,因为exrudeHPMCAS(++170°C)需要高温,聚合物和药可降解情况可能会改变 基于最近实验室研究三种固态表面活性剂(pooxamer 188,polxament 407和TPGS)和一种药模型(itraconaJohn大学,皇后区,美国纽约Serajuddin在药厂工作32年,在Sanofi-Aventis、BMS和Novartis担任科学管理职务曾任Novartis药厂执行董事和美国药厂研发主管并主管Novartis科技论坛,超过125篇论文和书章, 并共同创用13项专利承认他的科学专业成就后,他当选为美国药剂师协会和美国药剂师协会研究员,并获得三项最高奖项,即2010年AAPS设计开发研究成绩奖、2014年AAPS制造科学工程研究成绩奖和2015年AAPSLipid药物提供杰出研究奖AAPS中多位领导Serajuddin曾任预拟焦点组(1994-96年)和药理和药物提供科(2001年)主席2016年获Ralph Shangraw纪念奖,这是国际药商理事会最高科学奖学研究高超教士StJohn大学在2019年授予他大学杰出成绩奖章。
text-align:cent;sqle=bnbn-defaultbn-lg'hrefsm.us/webnar/register/WN_hXyjY7gLAY2gxxxxxwH.Solanki、N.G.Patel、N.G.SerajuddinT.M.2021. surfactics对Itraazole-HPMCS固体散射效果指令提交药理学杂志
Amorphous solid dispersions (ASDs) are popular for enhancing the solubility and bioavailability of poorly water-soluble drugs.使用各种方法制作asds并出现新手法本审查更新概述制作asds技术物理稳定性是ASD关键质量属性,配方效果 [.]
DerBeitrag Amorphous solid dispersions (ASDs) are popular for enhancing the solubility and bioavailability of poorly water-soluble drugs.使用各种方法制作asds并出现新手法本审查更新概述制作asds技术物理稳定性是ASD关键质量属性,已讨论了配方、设备及流程变量的影响,并讨论了下游处理对ASD物理稳定性的影响。选择策略建议确定合适的制造方法,这些方法可能有助于开发满足物理稳定性a/p/struct/dowload全文本 : 
Commonly used polymers for ASD preparation
Polymer Tg(°C) Solubility in solvents Hydroxypropyl methylcellulose 175‒185 Water, ethanol:dichloromethane (1:1, 2:1), methyl acetate:methanol (1:1) Hydroxypropyl methylcellulose acetate succinate 100‒110 Caustic water, acetone, methanol, dichloromethane, chloroform Hydroxypropyl methylcellulose phthalate 133‒137 Water, acetone, ethyl acetate, methyl ethyl ketone, ethanol:dichloromethane (1:1) methanol, dichloromethane, tetrahydrofuran Polyvinylpyrrolidone 175‒180 Water, acetone, ethanol, methanol, ethyl acetate, methyl ethyl ketone, dichloromethane, tetrahydrofuran Polyvinylpyrrolidone/vinyl acetate 70‒110 Water, acetone, ethanol, methanol, ethyl acetate, methyl ethyl ketone, dichloromethane, tetrahydrofuran Polymethacrylates derivatives (Eudragit®-L100, S100) >150 Water (only L100), acetone, ethanol, methanol, ethanol:dichloromethane (1:1) Cellulose acetate phthalate 160‒170 Acetone, ethyl acetate, methyl ethyl ketone Soluplus® 72 water, acetone, ethanol, methanol, dichloromethane
Source: Pharmaceutical amorphous solid dispersion: A review of manufacturing strategies
Conclusions
A successful development of amorphous solid dispersion formulations depends on three primary factors: active pharmaceutical ingredient properties, stabilizing polymer, and processing technology.聚合物为稳定药态化提供基础和基本基础,过程提供将系统变换成非态式所需的能量进程有效性对生成、捕捉和保存无变形式至关紧要成功这些过程取决于处理时间和超饱和条件形成固化散射时产生。
尽管在1960年代初发现固化散射,但用固化散射概念解决溶解挑战数十年来一直有限,部分原因是缺乏商业上可行的处理技术。然而,过去20年在开发药用ASD产品方面取得了显著进展,因为我们对ASD系统及其制造技术的理解有了相当大的发展,除开发数项产品外,还产生数项商业产品。喷雾干燥和HME成为制药行业ASD准备的主体,而更新方法则不断添加到工具箱中,保证提高产品质量、生产率和/或提高性能。
DerBeitrag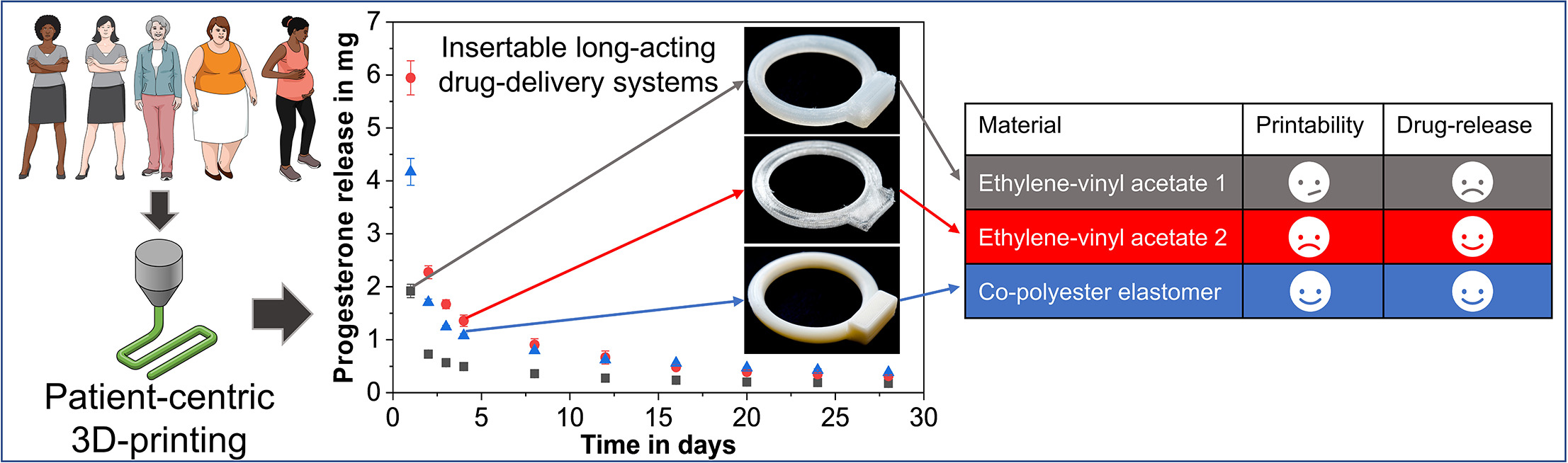
To improve patient compliance and personalised drug delivery, long-acting drug delivery devices (LADDDs), such as implants and inserts, greatly benefit from a customisation in their shape through the emerging 3D-printing technology, since their production usually follows a one-size-fits-most approach.三维打印LADDs使用受限制,但主要受三维可打印应用不足的限制 [.]
Der Beitrag To improve patient compliance and personalised drug delivery, long-acting drug delivery devices (LADDDs), such as implants and inserts, greatly benefit from a customisation in their shape through the emerging 3D-printing technology, since their production usually follows a one-size-fits-most approach.The use of 3D-printing for LADDDs, however, is mainly limited by the shortage of flawlessly 3D-printable, yet biocompatible materials. A novel 3D-printing excipient for long-acting drug delivery devices is introduced. A mechanistic understanding of material requirements for 3D-printing is presented. A novel progesterone immersion strategy for 3D-printed devices is developed. The novel excipient shows comparable drug permeability to ethylene-vinyl acetate. The present study tackles this issue by introducing a novel, non-biodegradable material, namely a polyester-based thermoplastic elastomer (TPC) – a multi-block copolymer containing alternating semi-crystalline polybutylene terephthalate hard segments and poly-ether-terephthalate amorphous soft segments.下一步详细描述材料3D可打印性,通过机械、风学和热学分析发现优于传统聚合物(ene-evenyacates),本研究首次建立对progeTPC展示了广选移植尺寸和复杂地理特征的3D可打印性。 
Highlights
Der Beitrag Novel polyester-based thermoplastic elastomers for 3D-printed long-acting drug delivery applications erschien zuerst auf Pharma Excipients.
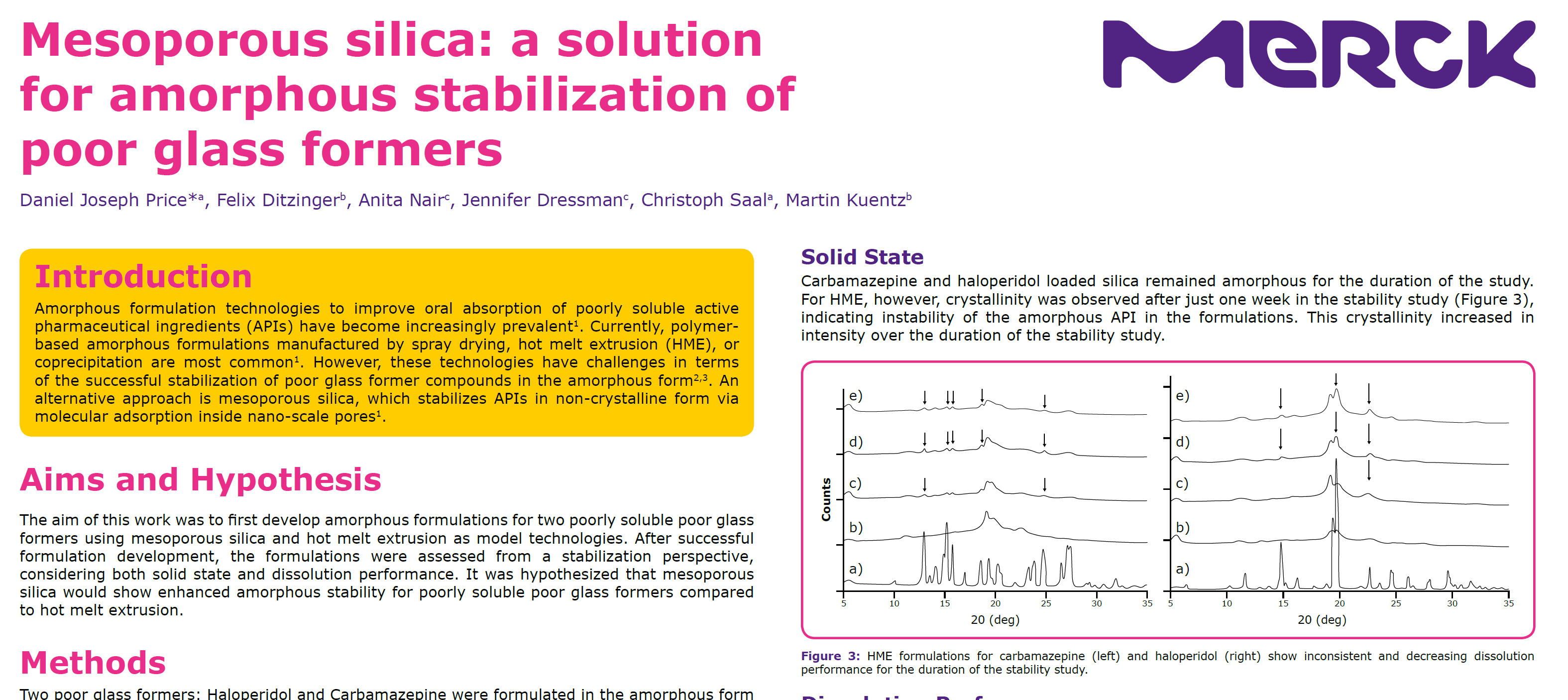
Introduction: Amorphous formulation technologies to improve oral absorption of poorly soluble active pharmaceutical ingredients (APIs) have become increasingly prevalent.聚合物非态配方目前最常用的是喷干热熔化或混合降水但这些技术挑战在于成功稳定s/www.pharmaexccidents.com/news/mesoporous-silica-poor-slipss/

Introduction: Amorphous formulation technologies to improve oral absorption of poorly soluble active pharmaceutical ingredients (APIs) have become increasingly prevalent.聚合物非态配方目前最常用的是喷干热熔化或混合降水但这些技术在成功稳定穷玻璃原复合物方面有挑战,偏差硅状单片解析法稳定非晶状单片解析纳米尺度内
 Download the full poster as a PDF here
Download the full poster as a PDF here
Conclusions: Although polymeric-based amorphous solid dispersions are most prevalent, they may not be suitable for poorly soluble compounds that are also poor glass formers.显示溶性差和无变稳定性差的这些复合物对配制典型聚合基技术具有挑战性,因为可能分解和内分解归根结底,这些复合物在开发药剂期间可能增加失效风险,因为它们从生物可用性和不定稳定性角度构成风险在这次研究中,我们证明不良玻璃组成a相形之下,多思西里卡显示为此类API提供最优稳定化Therefore, mesoporous silica could be an attractive formulation technology to expand the formulation toolbox for APIs that are poor glass formers.
Article information: Daniel Joseph Price, Felix Ditzinger, Anita Nair, Jennifer Dressman, Christoph Saal, Martin Kuentz
Der Beitrag Mesoporous silica: a solution for amorphous stabilization of poor glass formers erschien zuerst auf Pharma Excipients.

Crystallinity in an amorphous solid dispersion (ASD) may negatively impact dissolution performance by causing lost solubility advantage and/or seeding crystal growth leading to desupersaturation.研究的目标是评价Biculamide/Polyvinil-crystality内残留晶体化生成ASDss

Crystallinity in an amorphous solid dispersion (ASD) may negatively impact dissolution performance by causing lost solubility advantage and/or seeding crystal growth leading to desupersaturation.研究的目的是评价Bicalutamide/Polyvinyrli内射光谱学、极分光显微镜学和扫描电子显微镜学用来描述晶体动能和机制特征完全不动asd(0%晶度)不完全分解并结晶化为元可变多态(2形)非沉入条件下实现更高程度超饱和度,因为核分离前初始解密畅通性 。
ACSD内含剩余晶性显著降低超饱和度固介晶化(矩阵结晶化)耗尽非态固态,生长稳定多态水槽条件下完全不动asd和晶体混合比含剩余晶性assds快速释放后一系统矩阵结晶化导出高聚化晶体并具有高相对面积求解介晶化不是集中损耗的重要驱动力,因为当PVA出现求解时晶化增速慢BCL/PVAACDs残留晶化高风险来源于(1)晶子快速矩阵晶化传播和(2)稳定晶形生长这项研究对含残留晶性ASD性能分解有影响springer.com/article/10.1208/s12248-021-00598-6i>APSJ 23 23> 69>2021https://doi.org/10.1208/s12248-021-00598-6
Der Beitrag Amorphous Solid Dispersions Containing Residual Crystallinity: Competition Between Dissolution and Matrix Crystallization erschien zuerst auf Pharma Excipients.
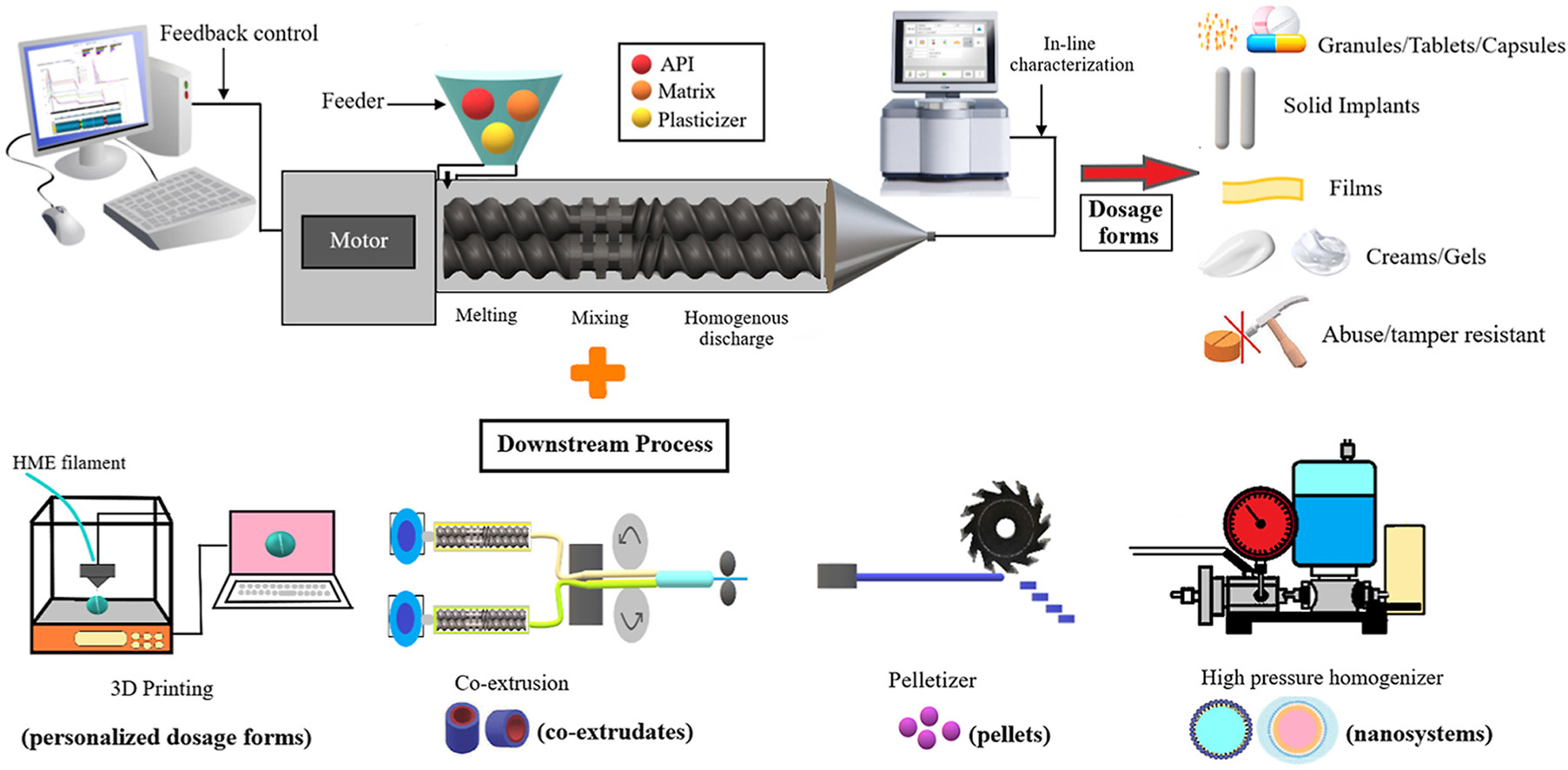
Hot-melt extrusion (HME) has emerged as a pioneering manufacturing technology for pharmaceutical industries, and the utilization is overgrowing due to its unit operation, cost-effectiveness, solvent-free nature, and continuous manufacturing as compared to conventional techniques.首要重心应放在研究配方和过程参数上,以满足需求。修改设备 [.]
Der Beitrag

Hot-melt extrusion (HME) has emerged as a pioneering manufacturing technology for pharmaceutical industries, and the utilization is overgrowing due to its unit operation, cost-effectiveness, solvent-free nature, and continuous manufacturing as compared to conventional techniques.首要重心应放在研究配方和过程参数上,以满足需求。By modifying the equipment design and varying a few processing conditions, a myriad of different dosage forms for various applications can be developed.
Highlights
This paper describes the Hot-melt extrusion (HME) equipment, process and materials.
Recent advancements in the applications of HME are reviewed.
Opportunities for HME technology in the veterinary field are discussed.
Recent patents of last five years in field of HME are presented.
An insight on pharmaceutical HME market globally is elucidated.
In-depth pre-formulation research is required for the rational selection of an active pharmaceutical ingredient, carrier, and additives for the smooth and continuous functioning of the HME process to achieve the desired product.近些年来,审查渠道从全局角度分析几种设备组件、处理需求、HME使用的材料和各种药送系统使用HME综合论文介绍数大新契机和创新方法,如三维打印剂量表、滥用确定式配方、共发药、时代药交付、联播器、纳米技术、半固片、双片粒子和多项应用。
文章还简要洞察2016至2024年间HME制药市场的全球状况Latterst,本审查汇总了最近四年HME数项应用,用表格方式提供药物,方便阅读文章试图为在这一领域工作的公式论者打下书目基础并推广这一领域的深入调查。 s/www.sciencedent.com/science/article/abs/pii/S1773224721001325热熔溢出:强调药方应用的最新进展,《药品科技杂志》,第63卷,2021年https://doi.org/10.1016/j.jddst.2021.102452.Der Beitrag Hot-melt extrusion: Highlighting recent advances in pharmaceutical applications erschien zuerst auf Pharma Excipients.

While hot melt extrusion was developed in the 1930s and initially used in the plastics and food sector, the pharmaceutical industry has now firmly embraced this technology platform in its manufacturing as a way of solving formulation challenges, particularly those around solubility enhancement.方法的优点包括使用广度 [.]
>Der Betrag

While hot melt extrusion was developed in the 1930s and initially used in the plastics and food sector, the pharmaceutical industry has now firmly embraced this technology platform in its manufacturing as a way of solving formulation challenges, particularly those around solubility enhancement.
Advantages of the approach include the ability to use a wide range of excipients and the ease with which process analytical technology can be incorporated.
In this podcast, we explore how HME has developed as proven technology for bioavailability enhancement of poorly soluble active pharmaceutical ingredients, and how it stacks up against other formulation options in terms of cost and scalability in order to deliver high quality, commercialised pharmaceutical products.
Source: Podcast recorded at CPhi discover with Bruce Frank
Vice President, Operations and Client Services
Lubrizol Life Science Health
Der Beitrag Podcast: Hot Melt Extrusion – Formulation Approaches and Technologies erschien zuerst auf Pharma Excipients.

In twin screw melt granulation, granules are produced by passing mixtures of drug substances and polymeric binders through twin screw extruder such that temperatures are maintained below melting point of drugs but above glass transition of polymers used, whereby the polymers coat surfaces of drug particles and cause their agglomeration into granules.自各种配方 [.]
Der Beitrag

In twin screw melt granulation, granules are produced by passing mixtures of drug substances and polymeric binders through twin screw extruder such that temperatures are maintained below melting point of drugs but above glass transition of polymers used, whereby the polymers coat surfaces of drug particles and cause their agglomeration into granules.
Since various formulation factors, such as binder type and concentration, and processing variables like extrusion temperature, screw configuration, and screw speed, can influence the granulation process, the present investigation was undertaken to study their effects on tabletability of granules produced.三种聚合物绑定器,/p>/d进程变量包括螺旋配置:eforminHCl低中高剪切值和actaminophen中低中剪切值摄取速率:20和60g/min螺旋速度100和300RPM构件和过程变量对可分片性有重大影响目标抗拉强度2兆帕可用所有聚合物并用所有处理温度求得,当Metformin HCl在180摄氏度和acetaminophen在130摄氏度被粒化时在其他温度下,目标抗拉强度在某些特定处理条件下可实现href="https://link.sprianger.com/aclll1228s122490211-02018-6jjects='blank'relshttps://doi.org/10.1208/s12249-021-02018-6
Der Beitrag The Effect of Process Variables and Binder Concentration on Tabletability of Metformin Hydrochloride and Acetaminophen Granules Produced by Twin Screw Melt Granulation with Different Polymeric Binders erschien zuerst auf Pharma Excipients.

Kollicoat® Smartseal (Methyl methacrylate and diethylaminoethyl methacrylate copolymer dispersion) which is used for taste masking and moisture protection, has shown the ability for forming solid dispersion.本研究旨在比照高熔点模型Itraconahrefs/www.pharmaexccepties.com/news/carrier-biopity-itraconazole/

Kollicoat® Smartseal (Methyl methacrylate and diethylaminoethyl methacrylate copolymer dispersion) which is used for taste masking and moisture protection, has shown the ability for forming solid dispersion.本研究旨在比照高熔点像Itraconazole模型研究in-vihrefs/www.pharmaex接收者.com/hot-melt-extrusi因此,拥有承运人可解决所有这些问题,将使业界为开发配方多选承运人。因此,作者试图解决这一问题并证明小承运人开发HME技术的能力Itraconadole固态散射编译、标注并测试大鼠的in-vivo 性能结果表明载波温度范围为120°C-220°C,行为良好Itraconazole对载波产生整形效果invitro 结果显示20倍溶性高于平面itraconazole.
hrefs/s/www.pharmaexsubjects.com/product/kolicoat-smartsal-30-d/science/abs/pi探索新载体提高Itraconazole生物可用性:热熔扩展固化分布https://doi.org/10.1016/j.jddst.2021.102541.
Der Beitrag Exploring novel carrier for improving bioavailability of Itraconazole: Solid dispersion through hot-melt extrusion erschien zuerst auf Pharma Excipients.

Vitamin B12 deficiency is associated with high risks of cognitive decline, neuropathy, megaloblastic anaemia and depression.因此,需要开发维他命B12创新配方恢复最优血清维他命B12水平(200-900皮克/毫升),特别是在复发病人中小说cyanobalin加载或分解薄膜开发 [.]
Der Beitrag

Vitamin B12 deficiency is associated with high risks of cognitive decline, neuropathy, megaloblastic anaemia and depression.因此,需要开发维他命B12创新配方恢复最优血清维他命B12水平(200-900皮克/毫升),特别是在复发病人中The present investigation deals with developing novel cyanocobalamin-loaded orodispersible films (ODFs) using Soluplus® as a film-forming polymer by hot-melt extrusion technology and application of Box-Behnken statistical design for formulation and process optimization.
Highlights
Robust, patient-friendly cyanocobalamin orodispersible films (ODFs) was developed.
Hot-melt extrusion technology was employed.
Soluplus® as film former was explored.
Quality by design was applied to optimize cyanocobalamin ODFs.
Pharmacokinetic studies of developed and marketed films were investigated.
The amount of Soluplus®, glycerine and menthol were the critical formulation variables influencing critical quality attributes of films such as tensile strength, disintegration time, dissolution and permeation rate.受物理化学特征测试时从熔膜中获取满意结果所有熔膜展示立即分解并完全分解 10分钟内完全分解ex-Vio渗透研究显示,比普通药解法更能从熔膜中渗透cyanobalinSEM和AFM分析显示,青歧蚁分布于优化膜全表层Raman光谱学确认HME处理后cyanobalin的稳定性。
a兔子模型应用来确定ODFs药效学并使用LC-MS/MS生物解析法营销Quicobaliveobalin从优化ODFs和营销产品生物利用率显示相似等离子浓缩时间剖面稳定性研究显示六个月内没有明显的物理变化science/artist.com/abs/pii/S1773224722392目标s='blank'rel='noopener'>阅读此文章Der Beitrag Development, optimization and in-vivo evaluation of cyanocobalamin loaded orodispersible films using hot-melt extrusion technology: A quality by design (QbD) approach erschien zuerst auf Pharma Excipients.

Interest in 3D printing for pharmaceutical applications has increased in recent years.与其他三维打印技术相比,热熔化叠加模HME技术可与FDM3D打印相伴并发并发关键药聚合物 uncls/www.pharmaexsubjects.com/3d-printing/coupling-hot-mel-fuse-depose/

Interest in 3D printing for pharmaceutical applications has increased in recent years.与其他三维打印技术相比,

